*This website was produced as an assignment for an undergraduate course at Davidson College.*
Orthologs of Nerve Growth Factor
Introduction:
Orthologs are defined as “genes found in different species, and whose divergence we can trace to the speciation events that gave rise to various species” (Sadava et al., 2008). An investigation of orthology, a type of homology, can give insight into gene evolution. Molecular evolution is, in turn, used to study protein function and how the function has changed over time.
To review material about the structure-function relationship of Nerve Growth Factor (NGF), follow this link to "My Favorite Protein: NGF" page.
Evolution:
NGF is one of the members of the neurotrophin family. Other members include brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), neurotrophin-4/5 (NT-4/5), and neurotrophin-6, and the evolution of these neurotrophins is poorly understood. In a particular study, one neurotrophin was isolated from Lampetra fluviatilis (river lamprey), and one was isolated from Myxine glutiosa (Atlantic hagfish). Studying these neurotrophins has allowed biologists to phylogentically analyze the neurotrophin gene family (Hallbook, 1999). The lamphrey and hagfish (jawless fish) diverged about 460 million years ago. In this study, neither of the two isolated neurotrophins resembled the known neurotrophins, and based on these findings, it is hypothesized that NGF (and other neurotrophins) was formed from duplication events from an intermediate, ancestral neurotrophin gene (Hallbook, 1999). A duplication event involves the copying of a gene, potentially altering its function. In essense, these duplication events likely took place in vertebrate history before the split of cartilaginous fish, but after the split of jawless fish.
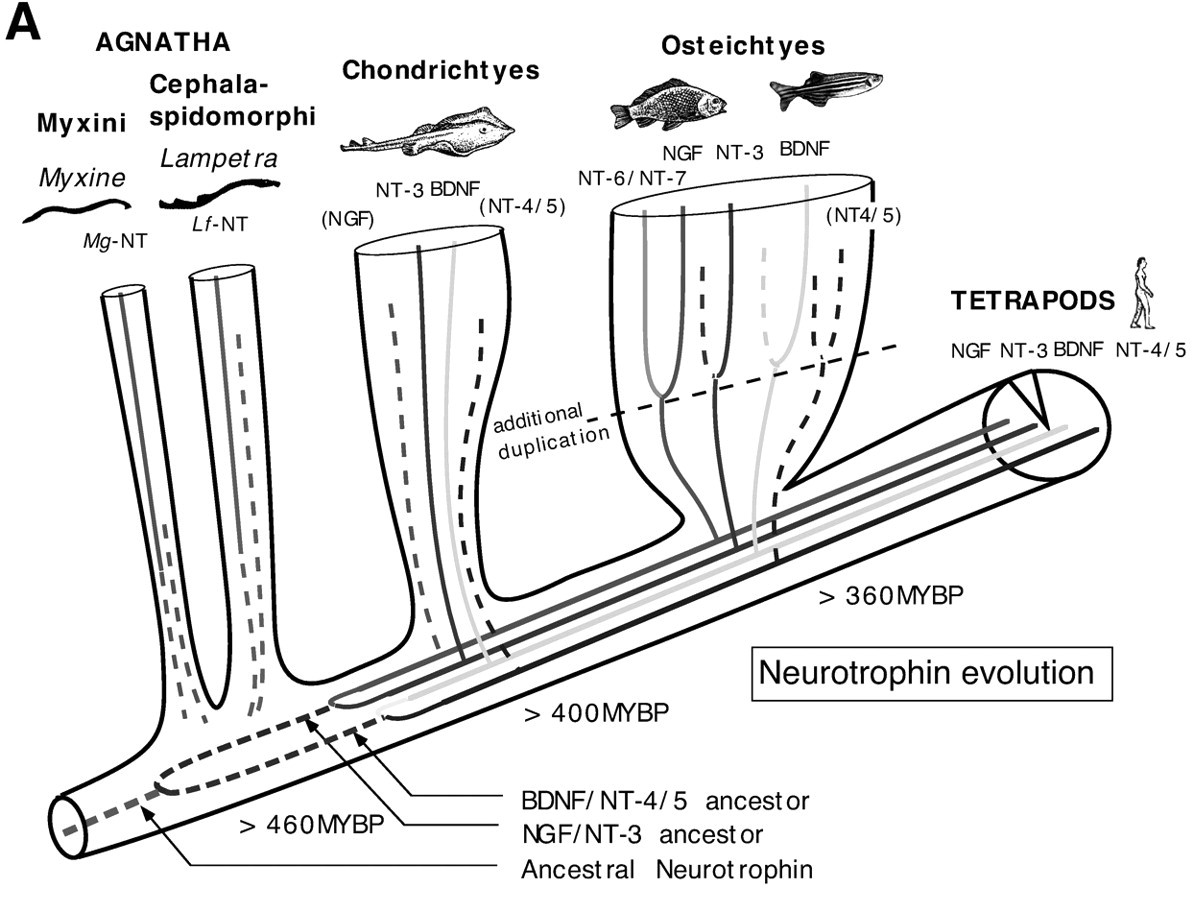
Figure 1. Depiction of neurotrophic evolution, illustrating divergence of branches of jawless fish (Agnatha), cartilage fish (Chondrichtyes), and bony fish (Osteichtyes). Observe the duplications events (line forks) that gave rise to NGF.
Image from Hallbook et al., 1998. Permission pending.
Mouse:
Nerve Growth Factor (NGF) was first isolated about 30 years ago from M. musculus (mouse) submaxillary gland. Although NGF is found in many vertebrate species, its relative abundance in this gland and in the venom of three families of poisonous snakes has prompted NGF to be studied most extensively among these species (Bradshaw, 1978). Other studies have investigated NGF from human, rat, chicken, viper, salmon, ray, and Xenopus (Hallbook et al., 1991). The sequence of one of the three subunits of mouse NGF, the β subunit, has been determined and studied intensively. The resulting amino acid sequence is depicted below.
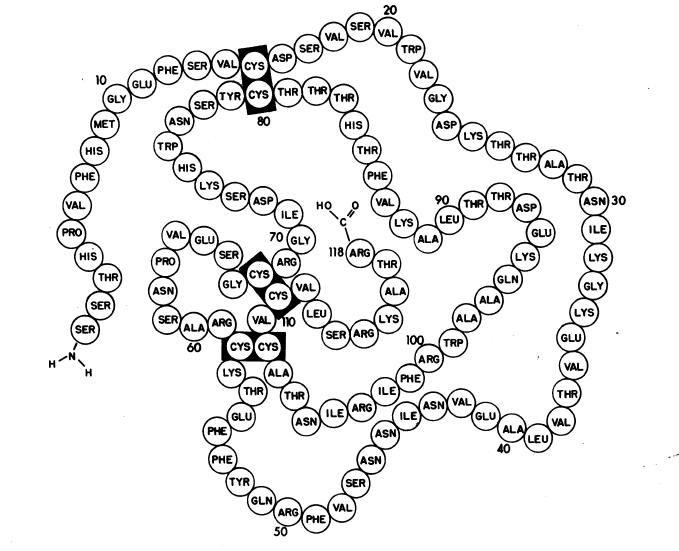
Figure 2. Representation of subunit of mouse NGF.
Image from Angeletti et al., 1971. Permission pending.
Performing a search on BLAST can give insight into possible orthologs. Some results, using the amino acid sequence of mouse NGF, included the following. Click on the link to view the findings.
R. norvegicus (Norway rat)
P. campbelli (Campbell's desert hamster)
M. natalensis (African soft-furred rat)
Human:
cDNA of the mouse β-NGF sequence has been used to isolate the gene for human β-NGF. This was possible because the coding regions in human β-NGF were highly homologous to the nucleotide and amino acid sequences in mouse β-NGF. A comparison of mouse and human β-NGF nucleotide sequences shows a high degree of homology, about 86% (Ullrich et al., 1983). Specifically, there is a large percentage of conservation within the cysteine residues of the mature NGF sequences; these residues are important in the formation of tertiary structure. This finding has led biologists to believe that the protein is highly conserved within mammals. Further homology has been demonstrated in studies using E. coli to express the human β-NGF sequence and immunological detection utilizing anti-mouse β-NGF antiserum (Ullrich et al., 1983).

Figure 3. Human β-NGF cDNA sequence.
Image from Borsani et al., 1990. Permission pending.
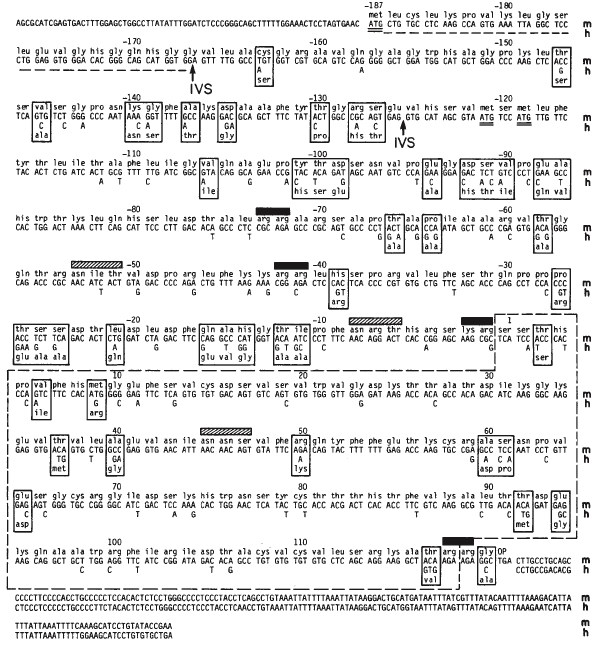
Figure 4. Comparison of mouse and human β-NGF cDNA sequences. The mouse coding strand (m) is shown entirely; differences in corresponding human sequences (h) are shown below. The boxes indicate regions at which nucleotide differences cause a change in the amino acid.
Image from Ullrich et al., 1983. Permission pending.
Other Genomic Species:
A search for NGF orthologs in other fundamental genomic species included C. elegans, Drosophila, Arabidopsis, yeast, and E. coli. No information was found concerning Arabidopsis, yeast, or E. coli. In addition, a search for orthologs of NGF found in C. elegans does not produce any notable results. However, one paper does note that vertebrate features have been lost from C. elegans, a possible explanation as to why no orthologs are found (Ruvkun, 1998).
It has been thought for a long time that NGF is absent from the Drosophila genome. However, a fairly recent study stated that neutrophic interactions do, in fact, occur in Drosophila. This group of biologists located a neurotrophic factor (DNT1) that they claim, with "compelling evidence," to be structurally related to known neurotrophins and conserved in insects. By varying the concentration of DNT1, the biologists were able to investigate the function of this neurotrophin. These findings imply that there may have existed a neurotrophin in the bilateral organisms' common ancestor, and this work has provided the opportunity to study neurotrophic function and related diseases more effectively (Zhu et al., 2008).
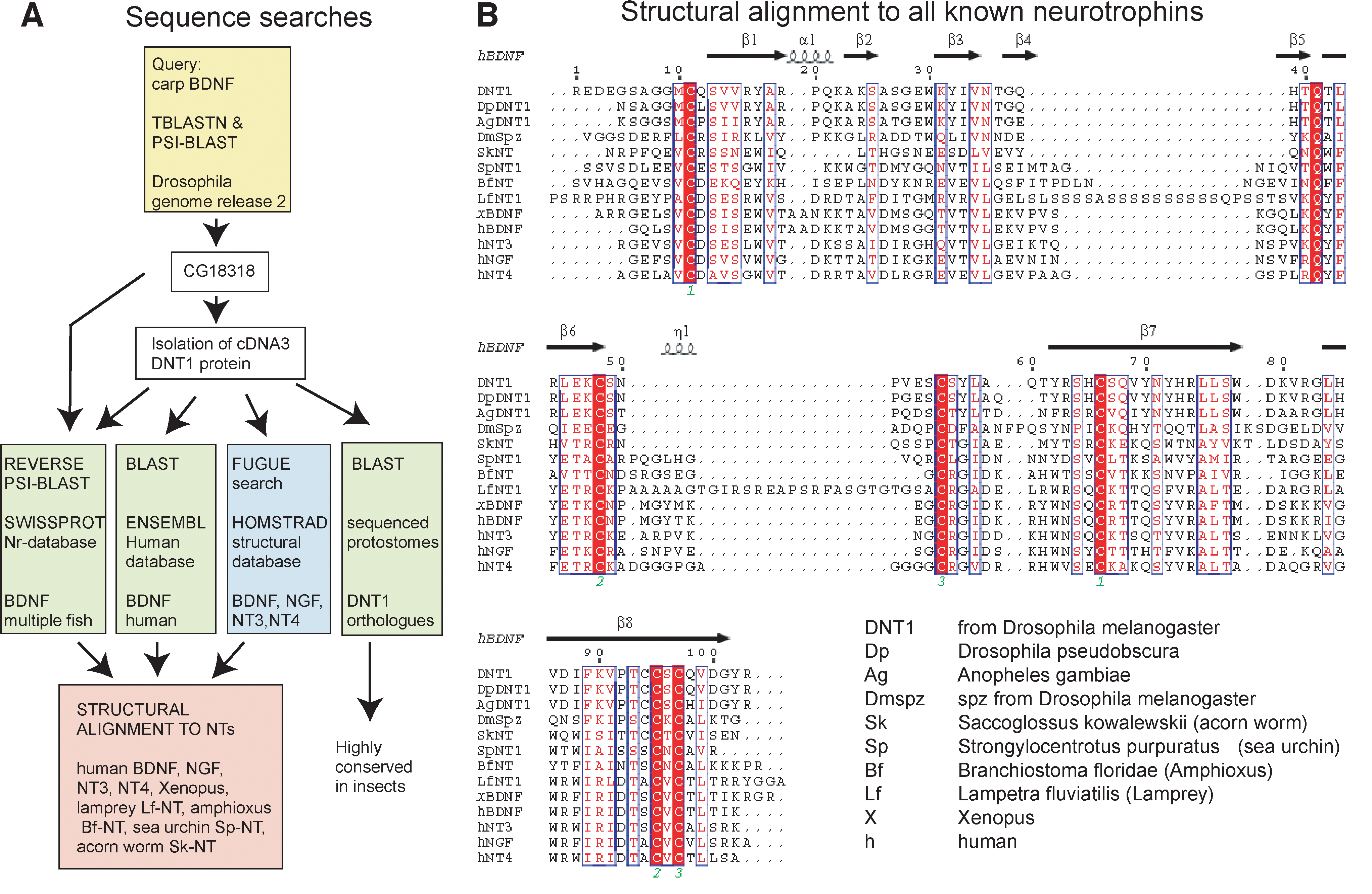
Figure 5. (a) Depiction of the sequence searches used to identify DNT1 (b) Alignments of all known neurotrophins with DNT1.
Image from Zhu et al., 2008. Permission pending.
Conclusion:
Because NGF plays a fundamental role in vertebrate nervous system development and maintanence, it is logical that it would be found mostly in vertebrates. NGF has mostly been studied from chordates, a major group that encompasses all vertebrates. It has been discovered in a variety of organisms, including amphibians, reptiles, fish, and mammals. It is highly conserved within these species, and most alterations have been found in variable regions (Hallbook et al., 1991).
In addition, the structural and functional similarities among members of the neurotrophin family itself suggest origination from a common ancestral gene (Hallbook et al., 1991). Studies have shown that the neurotrophin family and its respective receptors have co-evolved, allowing the formation of differing functions in various neuronal populations (Hallbook, 1999).

Figure 6. Nucleotide sequences of NGF isolated from various species. The dots represent identical bases.
Image from Hallbook et al., 1991. Permission pending.
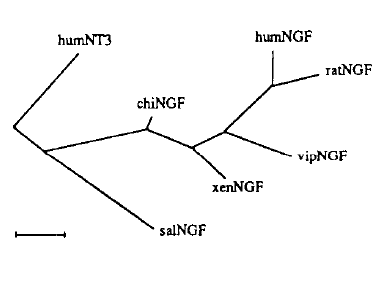
Figure 7. Deduced phylogentic tree of speciation of NGF.
Image from Hallbook et al., 1991. Permission pending.
References:
Angeletti R, Bradshaw R. 1971. Nerve growth factor from mouse submaxillary gland: amino acid sequence. Proceedings of the National Academy of Science 68: 2417-2420.
http://www.pnas.org/content/68/10/2417.full.pdf
Borsani G, Pizzuti A, Rugarli E, Falini A, Silani V, Sidoli A, Scarlato G, Baralle F. 1990. cDNA sequence of human β-NGF. Nucleic Acids Research 18: 4020.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC331136/pdf/nar00197-0321.pdf
Bradshaw R. 1978. Nerve growth factor. Annual Review of Biochemistry 47: 191-216.
http://arjournals.annualreviews.org/doi/pdf/10.1146/annurev.bi.47.070178.001203
Hallbook F, Ibanez C, Persson H. 1991. Evolutionary studies of the nerve growth factor family reveal a novel member abundantly expressed in xenopus ovary. Neuron 6: 845-858.
Hallbook F, Lundin L, Kullander K. 1998. Lampetra fluviatilis neurotrophin homolog, descendant of a neurotrophin ancestor, disloses the early molecular evolution of neurotrophins in the vertebrate subphylum. The Journal of Neuroscience 21: 8700-8711.
http://neuro.cjb.net/cgi/content/full/18/21/8700
Hallbook F. 1999. Evolution of the vertebrate neurotrophin and Trk receptor gene families. Current Opinion in Neurobiology 9: 616-621.
http://www.neuro.uu.se/mun/rtn/hallbook_currneuro.pdf
Ravkun G, Hobert O. 1998. The taxonomy of developmental control in Caenorhabditis elegans. Science 282: 2033-2041.
http://www.sciencemag.org/cgi/content/abstract/sci;282/5396/2033?maxtoshow=&hits=10&RESULTFORMAT=&fulltext=%28NGF+AND+ortholog+AND+in+AND+C.+AND+elegans%29&searchid=1&FIRSTINDEX=0&resourcetype=HWCIT
Sadava D, Heller C, Orians G, Purves W, Hillis D. Life: The Science of Biology 8th Edition. Massacheusetts and Virginia: Sinauer Associates Inc. and W.H. Freeman and Company, 2008. Print.
Ullrich A, Gray A, Berman C, Dull T. 1983. Human beta-nerve growth factor gene sequence highly homologous to that of mouse. Nature 303: 821-825.
http://www.nature.com/nature/journal/v303/n5920/pdf/303821a0.pdf
Zhu B, Pennack J, McQuilton P, Forero M, Mizuguchi K, Sutcliffe B, Gu C, Fenton J, Hidalgo A. 2008. Drosophila neurotrophins reveal a common mechanism for nervous system formation. PLoS Biology 6: 2476-2495.
http://www.plosbiology.org/article/info:doi%2F10.1371%2Fjournal.pbio.0060284
Davidson College Molecular Biology Homepage
Please send any questions, comments, or suggestions to moheinzelmann@davidson.edu.