This web page was produced as an assignment for an undergraduate course at Davidson College.
Alzheimer's disease (AD) is a form of dimentia that most commonly afflicts individuals late in life. AD symptoms include long-term memory loss, confusion, and irritability. The buildup of amyloid-beta (AB) molecules within the brain plays a major role in the pathogenesis of AD. Various types of AB species, including AB40, AB42, AB37, and AB38, are formed from the breakdown of amyloid precursor protein (APP) by initially beta-site APP-cleaving enzyme 1 (BACE1), and then by gamma-secretase. While AB42 is thought to contribute most heavily to the AD state, the other AB types may also play a role. Molecules that reduce APP breakdown to AB may be effective drugs against AD. Likewise, existing drugs or ingested compounds may increase AB buildup, therefore contributing to the disease state.
This page reviews the 2013 PLOS ONE article "The Proton-Pump Inhibitor Lansoprazole Enhances Amyloid Beta Production" (Badiola et al., 2013). The discussion includes a summary of the presented processes, results, and conclusions. Furthermore, summaries of the paper's figures provide a clearer identification of the data collected and the logical progression followed by the researchers. This review concludes with my response to the article as a whole.
In this article, Badiola et al. explored the effects of the FDA approved drug lansoprazole on APP breakdown to various AB species. Badiola et al. selected lansoprazol, a proton pump inhibitor (PPI) commonly used for gastric secretion inhibition, as a target drug for this project from previous research employing the therapeutic performance mapping system (TPMS). This previous work allowed for the identification of existing candidate drugs that could influence AD mechanisms. In vitro and in vivo experiments were conducted to draw conclusions regarding both the effects of lansoprazole on APP breakdown as well as its mechanism of action.
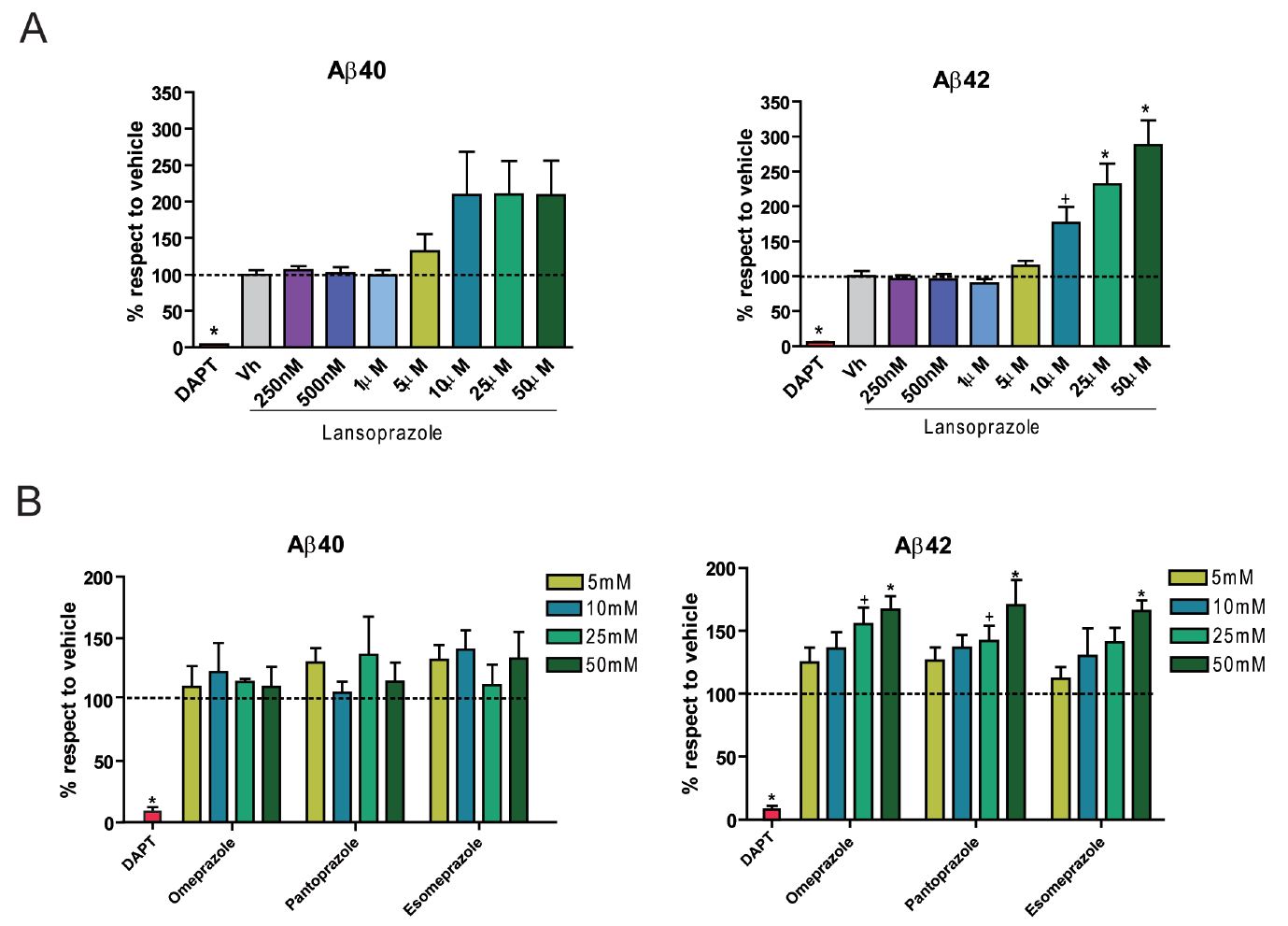
Chinese hamster ovary cell cultures transfected with human wt APP were used for in vitro experimentation. Results from ELISA immunoassays indicate that increasing concentrations of lansoprazole from 1-50 uM (for 24 hours) significantly increases AB42 levels (Figure 1A). Other tested PPIs, including omeprazole, pantoprazole, and esomeprazole, also resulted in increased AB42 production with increasing concentrations (Figure 1B). A non-significant increase in AB40 was also observed for all of the PPIs tested including lansoprazole. AB- immunoprecipitation follwed by MALDI mass spectroscopy allowed for quantitative identification of the concentration changes for the various AB species accompanying 50 uM lansoprazole addition (Figure 2A). The results indicated that lansoprazole acts as an inverse gamma-secretase modulator (iGSM), which increases AB42 concentrations and decreases AB38 concentrations through modulating gamma-secretase. Western blots suggested that lansoprazole also increases BACE1 activity. Furthermore, based on the MS results, Badiola et al. speculated that lansoprazole possibly increased activity of meprin beta metalloprotease. A hypothesized mechanism for the various enzyme activity increases was that lansoprazole induced a pH change within the system.
3xTg-AD transgenic mice were used for in vivo experimentation. Unlike with the in vitro experiment, results from ELISA assays from mice brain extracts demonstrated a significant increase in AB40 for both wt and transgenic mice treated with 100 kg/mg of lansoprazole. For both types of mice, AB42 did not significantly increase with the drug. This difference between the in vitro and in vivo results are attributed to the differences in the sampling techniques; the in vitro experiment only tested extracellular AB levels, while the in vivo experiment accounted for the intracellular AB as well.
After completing the study, Badiola et al.drew several conclusions.

A. This figure attempts to identify any AB level dependence on lansoprazole concentration. It reports in vitro results obtained from ELIZA immunoassays of AB40 and AB42 after treatment with lansoprazole. The samples were collected from the extracellular fluid of human wt APP tranfected Chinese hamster ovary cell cultures 24 hours after addition of the various lansoprazole concentrations. Vh represents vehicle alone without any lansoprazole. All AB percent increases are given with respect to the vehicle. DAPT represents a control without lansoprazole and with the addition of N-[N-(3,5-difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester (DAPT): a gamma-secretase inhibitor. As expected, the DAPT treated cells contain very diminished levels of both AB40 and AB42 because gamma-secretase was incapable of normal levels of function. Qualitatively, AB40 production appears to increase at concentrations at and above 10 uM. However, this observation is not quantitatively significant. AB42 does show a significant increase in production with increasing lansoprazole concentrations, showing that the drug does indeed have an effect.
B. This figure attempts to identify any AB level dependence on other PPIs. It reports in vitro results obtained from ELIZA immunoassays of AB40 and AB42 after treatment with PPIs other than lansoprazole. The samples were collected from the extracellular fluid of human wt APP tranfected Chinese hamter ovary cell cultures 24 hours after addition of the various PPI concentrations. All AB percent increases are given with respect to a vehicle alone control. DAPT represents a control without any PPIs and with the addition of N-[N-(3,5-difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester (DAPT): a gamma-secretase inhibitor. As expected, the DAPT treated cells contain very diminished levels of both AB40 and AB42 because gamma-secretase was incapable of normal levels of function. Again, results indicated no significant change in AB40 production but a significant increase in AB42 production with increasing concentrations of all three PPIs. This figure allows Badiola et al. to generalize the importance of their findings somewhat to other PPIs.
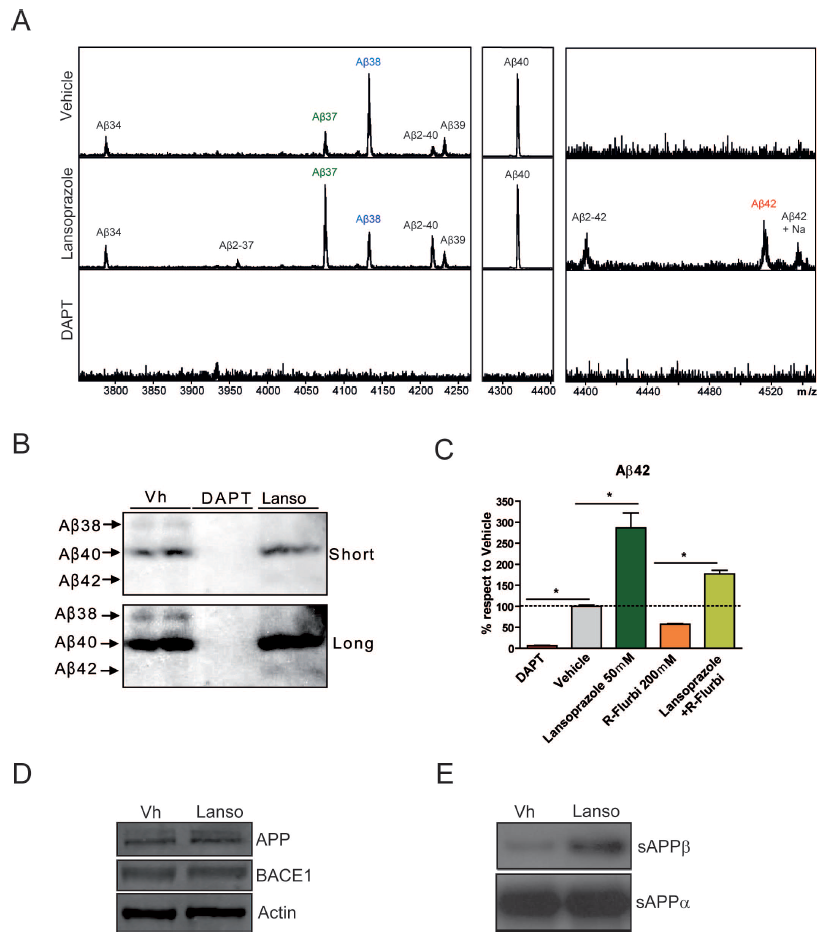
A. This figure attempts to identify specific changes in AB levels induced by lansoprazole treatment. Chinese hamster ovary cell cultures were prepared in the same way as discussed in Figure 1A and treated with 50 uM lansoprazole for 24 hours. The figure compares MALDI-MS results for the AB species obtained through immunoprecipitation from the extracellular fluid. A vehicle control is provided for comparison to the lansoprazole treated results. A DAPT treated control provides a proof of method as the lack of AB formation is expected. Results of the lansoprazole treated sample versus the vehicle control indicate an increase in AB42 and a decrease in AB38. This observed relationship is consistent with iGSMs, which interact with gamma-secretase to shift the cleavage sites of the enzyme. This provides one likely mechanism through which lansoprazole influences the AB system. Furthermore, it provides an explanation for the results obtained in Figure 1A. According to the text within the article, an increase in AB40 was also apparent from the MS results. However, the significance is not provided, y-axis magnitude is not provided, and this result would be inconclusive with those demonstrated in Figure 1A. Also worth noting is the appearance of AB2-X peaks, where X represents 40, 42, and 37. Badiola et al. hypothesize that these AB species emerge due to an increase in meprin beta metalloprotease activity. This seems plausible because this enzyme activity is pH correlated and lansoprazole is suspected of altering the pH.
B. This figure presents the relationship between AB38 and AB42 again through a different technique in order to verify the results. Using the same cell cultures, western blot analysis demonstrates the same results. Short and long indicate relative lengths of exposure. Again, DAPT appears as expected. The relative decrease in AB38 and the relative increase in AB42 can be seen. Again, from this relationship lansoprazole is thought to be an iGSM.
C. This figure analyzes the ability of lansoprazole to counteract the effects of a known AB42 lowering compound, R-flurbiprofen. Results were obtained from the same cell cultures by ELISA immunoassays. Treatment with DAPT, lansoprazole, and R-flurbiprofen by themselves provide the expected AB42 production changes. Results from the treatment with both lansoprazole and R-flurbiprofen indicate that lansoprazole can indeed significantly counteract the effects.
D. The iGSM activity of lansoprazole failed to identify the increase in AB40 and AB37. This figure demonstrates that the increase in those two species was not an artifact of either precursor (APP) or enzyme (BACE1) concentration variance between the vehicle treated control cultures and the lansoprazole cultures. Actin was used as a loading control. Because the band intensity appears the same, variations in the concentrations of APP and BACE1 do not account for the changes in AB37 and AB40 as shown in Figure 2A.
E. This figure attempts to identify the source of the increased AB40 and AB37 species since it does not appear to be caused by a concentration artifact or by gamma-secretase. sAPP-beta is the product produced from APP breakdown by BACE1. The increase in sAPP-beta from vehicle to lansoprazole treated indicates an increase in BACE1 activity. sAPP-alpha is the product formed when APP is broken down by alpha-secretase, whose activity appears unaffected. The increase in sAPP-beta provides an explanation for the increase in AB40 and AB37. Similarly to the enzyme meprin beta metalloprotease, the increase in BACE1 is speculated to be due to a lansoprazole induced pH variation.
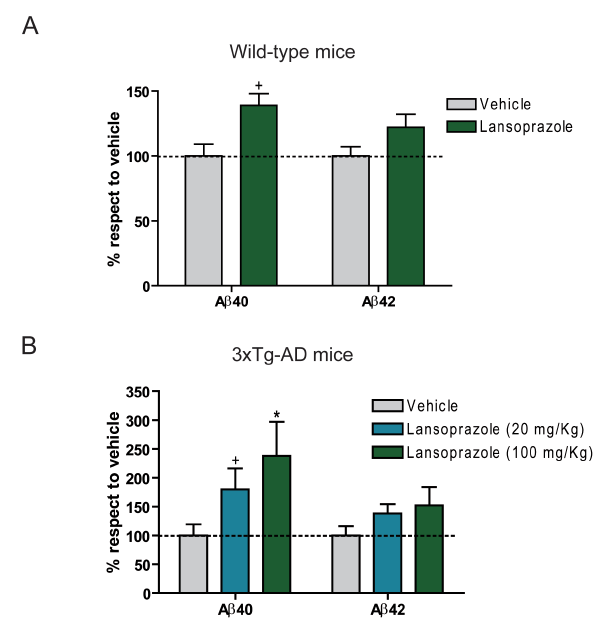
This figure identifies the in vivo AB40 and AB42 level dependence on lansoprazole treatment. Unlike the in vitro cell culture results from Figures 1and 2, this experiment provides results from wild-type and AD transgenic mice brains. The sample collection involes extraction kits and then ELISA assays. Also unlike the cell culture experiments, these experiments extracted both intracellular and extracellular AB species (which could account for in vitro/ in vivo data discrepancies). Furthermore, the in vivo results indicate that lansoprazole induced AB increases appear to occur even at relatively low concentrations (compared to concentrations used for its current drug functionality).
A. This figure illustrates data for wt mice treated with 100 mg/Kg lansoprazole (5 consecutive days). Contrary to the in vitro results, significant increase in AB40 is apparent compared to vehicle, while no significant change is observed for AB42. Regardless, an increase in AB is still observed.
B. This figure illustrates data for the AD transgenic mice treated with 20 mg/Kg or 100 mg/Kg of lansoprazole (5 consecutive days). Again, significant increase was observed for AB40, while no significant increase was seen for AB42. Interestingly, the increase of AB40 for the AD mice was greater than that for the wt mice. This could indicate a potential progression to the AD inductive effects of lansoprazole.
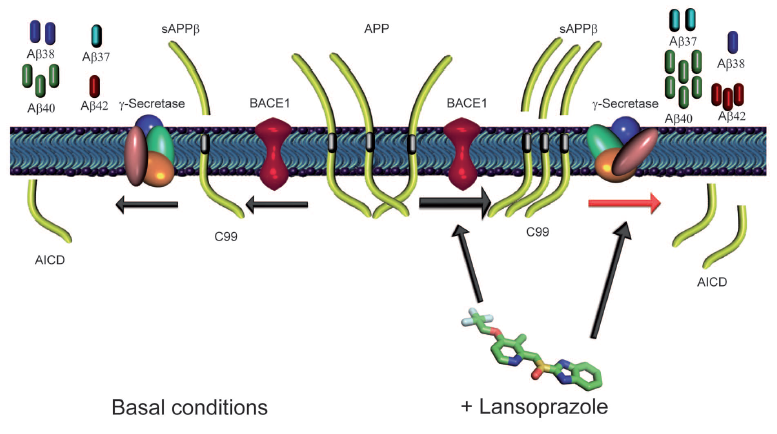
This figure attempts to summarize and connect the findings of this research. It depicts a general hypothetical mechanism of lansoprazole action on the production of AB. The left side is the wt basal conditions (normal) and the right represents the alterations made by lansoprazole. As previously demonstrated, lansoprazole influences BACE1 to increase the production of sAPPbeta compared to the basal conditions (increased BACE1 activity as illustrated by larger arrow). Relative amounts of product throughout the figure are illustrated qualitatively. Lansoprazole also influences gamma-secretase. Instead of upregulating the enzyme, it simply induces a shift in the cleavage patterns of sAPPbeta. When taken together, these result in the increse of AB40, AB37, AB42, and AICD (APP intracellular domain fragments) and the decrease in AB38 caompared to basal conditions. The AICD decrease was attributed to the increased relative concentrations of sAPPbeta compared to sAPPalpha (not illustrated).
Overall, the research presented in this paper seems well done. Badiola et al. clearly provide their rationale behind the various experiments and lead the reader through the results. The conclusions drawn are well qualified and don’t attempt to exaggerate what the data supports. As part of this, Badiola et al. clearly indicate some of the shortcomings within this study, which should be explored more fully in the future (i.e. unclear underlying mechanisms). Furthermore, the paper provides reasonable speculation regarding unexpected results. An example of this can be found in the discussion on the MALDI-MS data depicting AB2-X peaks. The researchers provide the reasonable explanation of lansoprazole induced pH changes influencing the protein often responsible for those cleavage products. The discussion regarding the apparent conflicting results for AB40 and AB42 lansoprazole dependence observed between the in vivo and in vitro experiments provides another example of reasonable speculation. Specifically, they discuss significant differences between the in vivo/ in vitro AB extraction techniques. Finally, I found the context with which the paper was framed very helpful. The concentrations used were specifically selected to be lower than the concentrations employed for the drug’s FDA approved treatment role. This makes the significance of the results apparent.
While the paper communicated the study very well for the most part, I do have certain issues with the presentation. A major issue was the general confusion involving AB40 results. In Figure 1A, the AB40 results indicate a lack of significant increase with lansoprazole treatment. On the other hand, the discussion on this figure and the MS results (Figure 2A) suggest that a significant increase did occur. This seems unverifiable due in part to the lack of a signal y-axis for the MS results. Along the same lines, western blot quantification would have been nice to see as a reader. Finally, because of the apparent specificity of the various AB species, a more in depth discussion on each would be a welcome addition to the introduction. Fortunately, these were the only major critiques I have for this paper. Again, because Badiola et al. did not overextend themselves and the collected data when drawing their carefully qualified conclusions, the end product seems an accurate depiction of what the study truely accomplished.
© Copyright 2013 Department of Biology, Davidson College, Davidson, NC 28036
Send comments, questions, and suggestions to: johimes@davidson.edu