This web page was produced as an assignment for an undergraduate course at Davidson College.
Lactobacillus plantarum Paper Summary
Summary
Investigators in the lab of Francois Leilier sought to determine how the microbiota regulated growth during undernutrition conditions in mice. Chronic undernutrition leads to a growth hormone resistance that keeps the mice from achieving normal weight and body length. Comparing wild type (WT) and germ free (GF) mice, the investigators were able to show that the microbiota acts through the somatotrophic axis to increase IGF-1 signalling, leading to normal mouse growth. The somatotrophic axis is part of the anterior pituitary that produces growth hormones. The GF mice did not have the same growth gains. The WT mice also grew more than GF mice in depleted feeding conditions. The investigators used several types of methods including measuring the gains in weight or body length, and also measuring levels of several players in the somatotrophic axis from the sera of mice. Additionally, the investigators wanted to determine if the L. plantarum bacterial strain was a necessary component in order for the microbiota to positively influence growth. Through mono-colonizing experiments, GF mice were cultured with one of two specific L. plantarum strains, and these mice had better overall growth and higher somatotrophic axis activity up to 56 days after birth.
Conclusions
These data as a whole establish the role of the microbiota in regulating the activity of the somatotrophic pathway. During breeding diets, WT mice featuring a normal gut microbiota grew more than GF mice. The microbiota additionally helped reduce otherwise stunted growth in mice during a depleted diet. The microbiota acts through the somatotrophic axis and IGF-1 signaling to systemically increase growth of key organs including bone length and mass gain. Colonizing GF mice with L. plantarum abrogated the stunted growth effects during depleted diet. Broadly, microbiota supplements of the L. plantarum bacterial strain could aid the effects of malnutrition on human growth.
Personal Critique
The investigators used a broad variety of
techniques that overall convinced me both of their methods and their
conclusions that the microbiota was one of the main causes of increased
growth in infant mice. Their text adequately explained the figures and the
color scheme and labeling system was consistent throughout the paper. The
conclusions for each data set were also clearly stated. One critique I do
have is in their organization. Figures 3 and 4 feature data from the
additional experiments to colonize specific L. plantarum strains
in GF mice, asking whether one strain over the other has a better effect
at increasing growth during depleted diet conditions. The text for this
part of the experiment was put at the end of the paper rather than with
the rest of the text corresponding to those figures and their methods.
This was rather confusing to me, that the paper essentially ignored half
of two of the figures until the very end of the paper. It would have been
better to introduce these experiments and the rationale behind them when
the figure came up in the paper. Otherwise, the conclusions were well
explained and the results were applied to bigger situations of human
infant malnutrition and possible treatments with microbiota and the L.
plantarum bacteria.

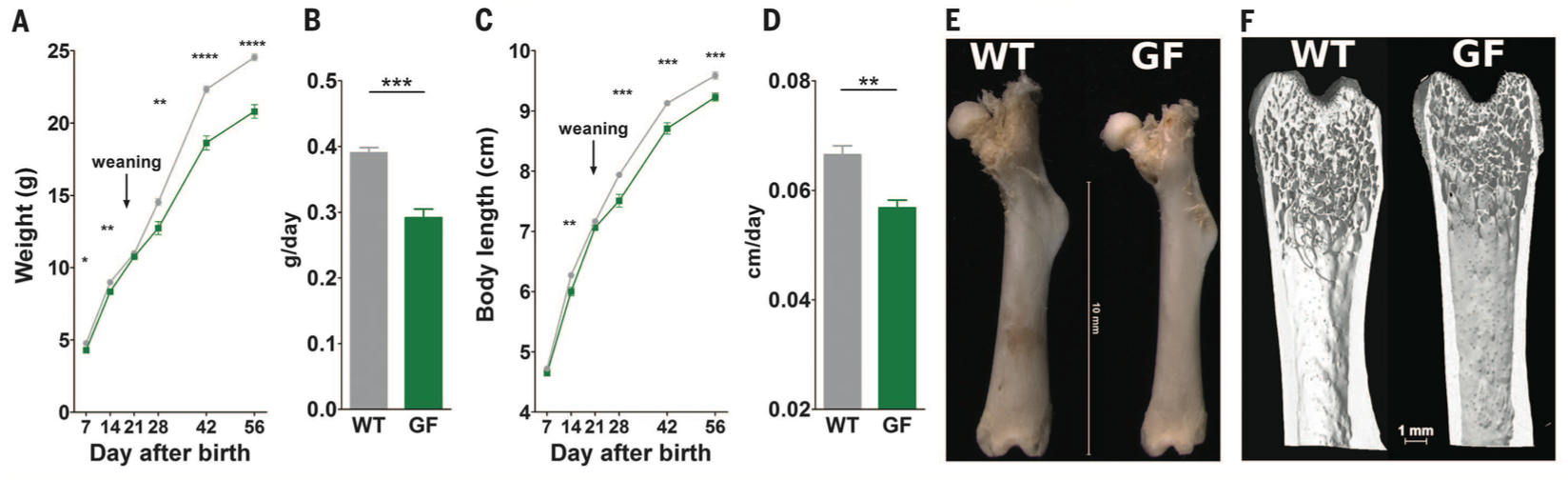
Figure 1. The data in figure 1 are a result of the investigators wondering whether the microbiota contributes to mouse growth in terms of their weight and length during normal feeding conditions. They used wild type (WT) and germ free (GF) and compared growth measurements. The wild type mice had a normal, unaltered microbiota, but the germ free mice did not have a microbiota, and thus any differences between the two types of mice could be attributed to microbiota interactions. Panels A and C show weight and length differences between the two strains of mice over a period from 7 to 56 days after birth. Panels B and D further break down growth into grams or cm per day (for weight and length respectively). The germ free mice weighed less and had shorter body length compared to the wild type mice. Panels E and F show representative femur bones at day 56 between the WT and GF mice. The bones of the GF mice are shorter, and panel F shows that they've developed less as well. Both WT and GF mice had the same adipocyte size and fat pad size, and so the increased weight gain in WT mice cannot be attributed to fat. Rather, the microbiota in WT mice is influencing growth of organs and is associated with systemic somatic growth.
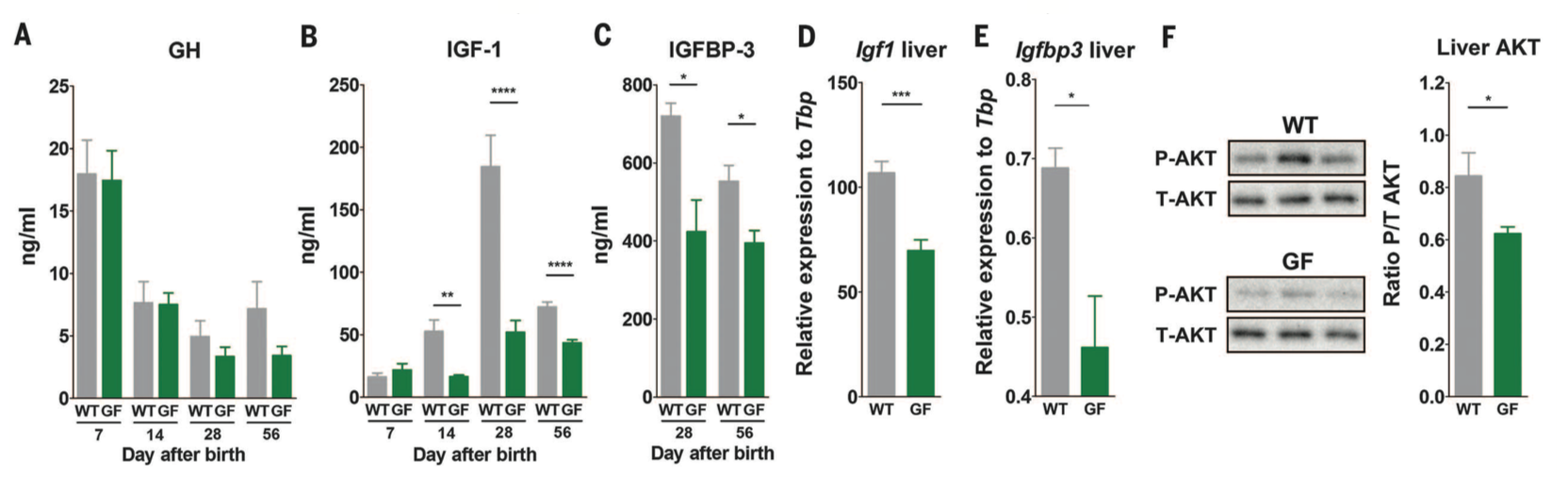
Figure 2. The next question the investigators wanted to answer was how the microbiota influenced growth. They measured the levels of the somatotrophic axis which drives systemic growth. Panels A-C measure levels of growth hormone, IGF-1 (induced by growth hormone), and the IGFBP-3 (IGF-1 binding protein-3) respectively in the sera of WT and GF mice. Growth hormone and IGF-1 were measured 7, 14, 28, and 56 days after birth. Levels of growth hormone were not significantly different at any time points between WT and GF mice, but the levels of IGF-1 were higher in WT mice at 14, 28, and 56 days. The IGF binding protein (IGFBP-3) was measured 28 and 56 days after birth, and also had higher levels in WT mice. These data suggest that the microbiota results in increased somatotrophic axis activity, through measuring important players involved in the signaling pathway. In panels D and E, the investigators show levels of Igf1 and Igfbp3 gene expression relative to a Tbp control in the liver specifically, and we find that again the somatotrophic axis is higher in WT mice.
Additionally, the investigators measured phosphorylated AKT. Phosphorylation of AKT at a specific serine is a marker of the IGF-1 receptor signaling activity, and therefore is another measure of somatotrophic axis activity. In panel F is shown a western blot measuring both phosphorylated and non-phosphorylated AKT levels in WT and GF mice. The western blots are quantified to the far right. The bands for P-AKT are darker in WT mice indicating greater levels of phosphorylated AKT, and therefore increased IGF activity. These data collectively confirm that the somatotrophic axis is more active in the presence of a microbiota, and this causes systemic growth in WT mice.
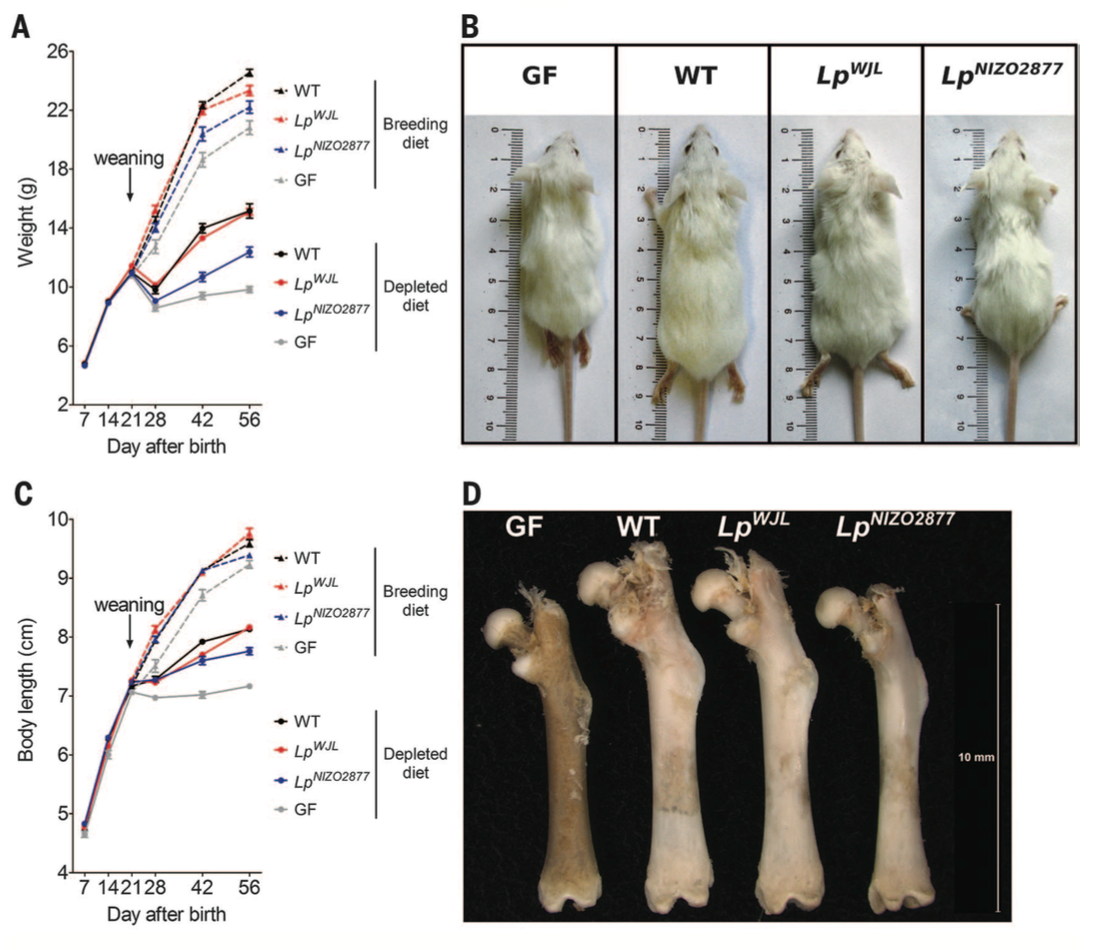
Figure 3. Now we know that the microbiota contributes to increased mouse growth through somatotrophic axis activity in mice during normal breeding feeding. The next question was how does the microbiota contribute in situations of under-nutrition. To answer this, investigators again measured weight and body length in WT and GF mice but in a breeding diet compared to a depleted diet. The weight of WT mice was higher than GF mice in the breeding and the depleted diet conditions (panel A). Similarly, WT mice had greater body length than GF in breeding and depleted feeding (panel B). The size of the both WT and GF mice was bigger in the breeding than depleted feeding conditions, but the microbiota in WT mice appeared to compensate for under-nutrition. There are no statistics shown here, so it is unclear whether these differences are significant, but the overall trend is that GF mice have stunted growth.
The investigators also wanted to know what specific bacterial strain contributed to this increased growth in WT mice. Research in Drosophila development had previously identified Lactobacillus plantarum strains as promoting growth capacities. The investigators therefore conducted mono-association experiments in GF mice. Essentially, they added two strains back to germ free mice and colonized them in the mice gut. These colonized mice were subjected to the same breeding and depleted diet, and measured for their weight and body length. The Lp-WJL strain aided mouse growth in both diet conditions; the Lp-NIZO2877 strain aided growth compared to the GF mice, but to a lesser extent than the former strain (panels A and C). Finally, the investigators measured the length of all four types of mice, pictured in panel B, and the length of their femurs (panel C).
These data collectively reveal that the microbiota aids growth in under-nutrition conditions, and that L. plantarum is a specific bacterial strain that prevent otherwise stunted growth in GF mice.
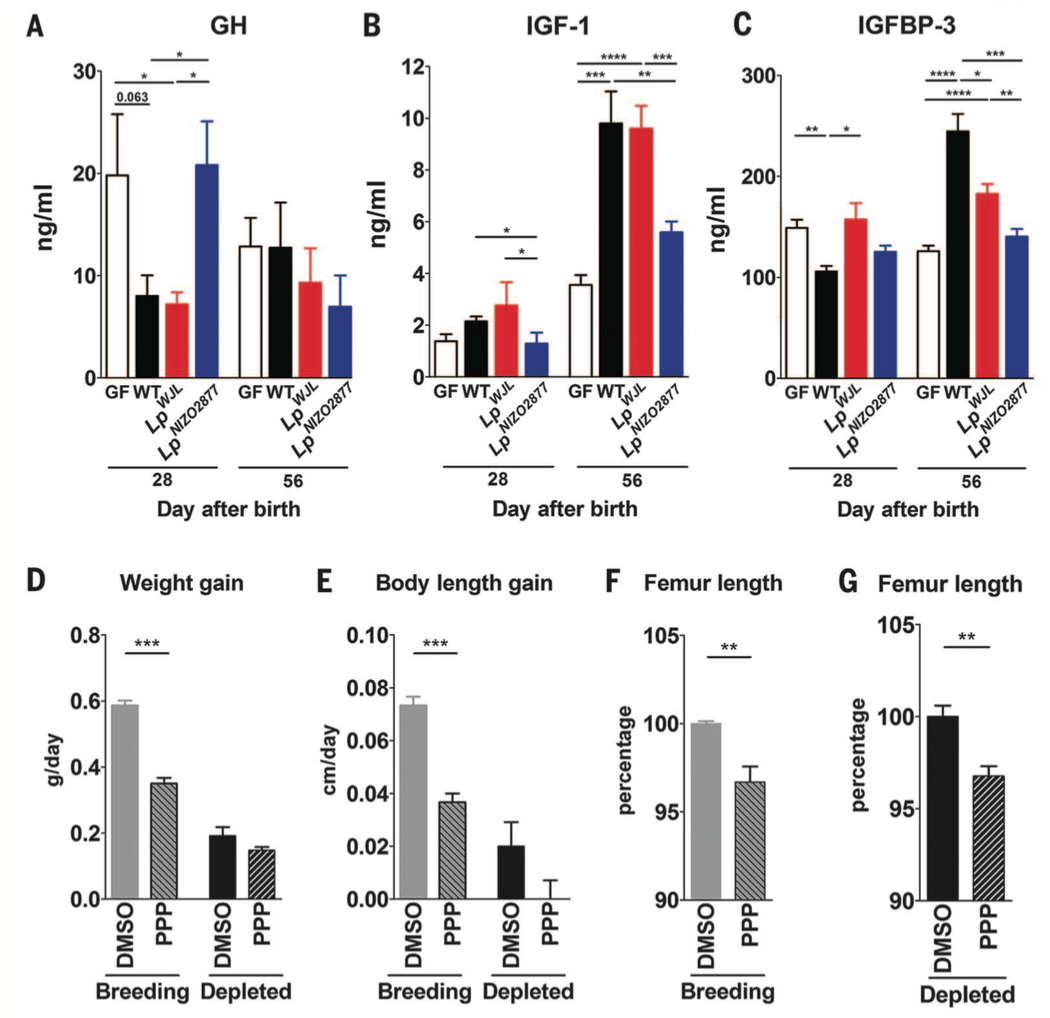
Figure 4. Similar to the experiments in Figure 2, the investigators repeated their measurements of elements of the somatotrophic axis in the depleted diet condition (panels A-C). There were no significant differences in growth hormone level between GF and WT mice 28 or 56 days after birth. However there were higher levels of IGF-1 in WT mice compared to GF mice 56 days after birth. The levels of IGFBP-3 started out higher in GF mice at 28 days, but by 56 days, the WT mice had significantly higher levels. When GF mice were supplemented with the Lp-NIZO2877, the growth hormone levels were comparable to GF mice at 28 days, but by 56 days there were no differences in growth hormone levels between any of the 4 mice groups. These data suggest some sort of growth hormone resistance as the mice age. The general trend for the IGF-1 levels was that L. plantarum supplemented GF mice had higher levels than the GF mice but lower levels than WT mice by 56 days. Between the two L. plantarum strains, the Lp-WJL increased IGF-1 levels to a greater extent. The data for IGFBP-3 levels followed a similar trend at 56 days: WT mice had higher levels, but the L. plantarum supplemented somatotrophic axis activity in GF mice.
Panels D-G of this figure show data for an inhibitor of the IGF receptor. When the receptor is inhibited with picropodophyllin (PPP), weight gain, body length, and femur length of WT mice are lower in both breeding and depleted diets compared to giving a DMSO control.These data further support the conclusion that the somatotrophic axis and IGF-1 signaling activity are contributing to system growth in mice, and that IGF-1 activity is essential.
Reference
Schwarzer, M., Kassem, M., Gilles, S., et al. (2016) Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition. Science 315(6275): 854-857.