This web page was produced as an assignment for an undergraduate course at Davidson College.
The Alzheimer's Transcriptome
Alzheimer's Disease and Alipoprotein E
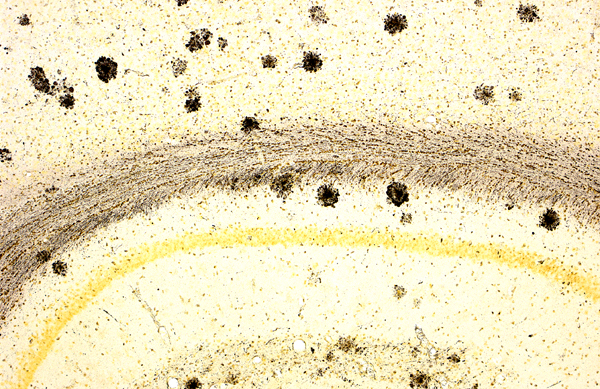
Alzheimer's Disease (AD) is a fatal neurodegenerative disease
characterized by progressive memory loss. The disease is believed to
be caused in part by the aggregation of insoluble fibrils of the
protein amyloid-beta. Amyloid-beta is a domain of the membrane protein
Amyloid Precursor Protein (APP), which is cleaved to release the
extracellular amyloid-beta protein component. The aggregated clumps of
amyloid-beta form amyloid plaques, a key histological hallmark of AD
(Huang 2012) (Figure 1).

Figure 1: Amyloid Plaque. Image courtesy of neurodigitech.com.
Alipoprotein (apo) E4 is an allele of the ApoE gene with a strong genetic link to the development of late-onset Alzheimer's Disease (LOAD). The exact functions of ApoE4 in AD development is a topic of intense debate and study. Some research shows that apoE may be involved in the removal of amyloid-beta deposits while other studies indicate amyloid-beta independent roles of ApoE in AD progression (Huang 2012). It is likely that ApoE4 plays a multifaceted role in human neurons, as it has been implicated in a variety of cellular processes (Huang 2012).
Gene Expression Changes in ApoE4 carriers
A 2013 study published in Nature
investigated whole-transcriptomes from healthy carriers of the ApoE4
allele and LOAD patients. In this study, gene expression in the cerebral
cortices of ApoE4 allele carriers and LOAD patients were compared to each
other and to a reference transcriptome (Rhinn 2013). The study found that
ApoE4 carriers exhibited changes in gene expression that resembled the
gene expression changes found in Alzhemier's patients. Additionally, the
researchers identified novel genes with altered expression in ApoE4
carriers and LOAD patients, including APBA2, RNF219,
and SV2A (Rhinn 2013).
Genomics as a Discovery Science
This project began with a rather open-ended and perhaps almost obvious hypothesis: the transcriptomes of ApoE4 allele carriers and LOAD patients would differ from the transcriptomes of healthy patients. As the topic of study was so broad, this research study is a good example of discovery science. In discovery science, large volumes of experimental data are analyzed to identify patterns or correlations. In the present study, entire transcriptomes were evaluated in the effort to find differences transcriptional between genotypes and phenotypes.
Despite the breadth of the
research study, researchers were able to reach very specific conclusions:
the discovery of precise genes with transcriptional differences in ApoE4
carriers and LOAD patients. This study is a wonderful example of how
genomic methodologies can allow for successful management of what would be
an otherwise overwhelming amount of raw experimental data.
Genomic Technology in This Study
This study measured gene expression through quantitative reverse transcription polymerase chain reaction, or qRT-PCR (Rhinn 2013). This method is a combination of two common techniques in molecular biology, reverse transcription PCR (RT-PCR) and real-time PCR (qPCR). In RT-PCR, RNA is reverse transcribed to complementary DNA via reverse transcriptase and amplified through PCR. In qPCR, PCR products are labeled with fluorescent probes during thermal cycling. As the PCR product is amplified, fluorescent signal accumulates at a rate that is proportional to the amount of starting DNA. In qRT-PCR, the processes are combined, allowing for the quantitative analysis of RNA.
In this study, qRT-PCR was
used to quantify the entire transcriptome of patients in the various
categories. This transcriptome-wide approach allowed for changes in total
transcription in the ApoE4 genotype or LOAD phenotype to be evaluated.
Take Home Message
This paper demonstrates how basic discovery science with genomics methodologies can be employed to make clinically relevant discoveries from immense data.
Evaluation of the Project
This paper begins with a
simple premise: the Alzheimer's transcriptome will bear considerable
differences than a healthy transcriptome. Despite this basic and simple
premise, the researchers are able to identify specific molecular
differences in the respective transcriptomes and identify novel genes in
ApoE4 signaling in Alzheimer's development. All in all, this was a very
effective research strategy that yielded powerful results through the
application of genomics methodologies.
References
Huang, Y. and L. Mucke. 2012. Alzheimer mechanisms and therapeutic strategies. Cell 148 (6): 1204-1222.
Pittcon. 2015. Understanding Alzheimer's Disease through genomics and proteomics [Internet]. [Cited 2016 Jan 22]. Available from http://www.news-medical.net/whitepaper/20151111/Understanding-Alzheimere28099s-Disease-through-genomics-and-proteomics.aspx.
Rhinn, H., Fujita, R., Qiang, L., Cheng, R., Lee, J. H., and A. Abeliovich. 2013. Integrative genomics identifies APOE e4 effectors in Alzheimer's Disease. Nature 500: 45-50.
Genomics
Page
Biology Home Page
© Copyright 2016 Department of Biology, Davidson College, Davidson, NC 28035