

Figure 1. These are images of real plasmids.
The images can be found at http://www.thermomicro.com/apps/lifesci/lifesci17.htm
The method to be analyzed appears in a paper by Gutierrez-Campos, Torres-Acosta, Saucedo-Arias, and Gomez-Lin titled, "The use of Cysteine protease inhibitors to engineer resistance against potyviruses in transgenic tobacco plants," Nature Biotechnology, Vol. 17. Dec. 1999.
Purpose of the method: Use techniques of plant genetic transformation to help plants develop resistance against viruses.
Why it should be used: The new method is cheaper and less protracted than conventional methods.
Background: In plants, resistance to viruses has been achieved
through a number of methods, including through transformation with natural
viral resistance genes, anti viral proteins, and sense and antisense RNA.
Pathogen derived resistance (PDR) has previously been used to induce resistance
to potyvirus infection. PDR involves transformation with genes derived
from the pathogen itself and is limited by a number of factors, including
the specificity for the viral strain from which the gene was isolated,
the large number of different potyviruses, and the genetic variation within
a particular potyvirus (Lomonossoff, 1995).
The authors propose a new method for controlling
potyviral infections. An essential step for the replication of potyviruses
is the activity of cysteine proteases. Cystatins (cysteine protease
inhibitors) are powerful and specific inhibitors of cysteine proteases,
and could provide a novel and specific method to control such infections.
Cystatins from corn and rice (oryzacystatin I) in transgenic plants have
shown to increase resistance against insect gut proteases and plant colonization
by the nematode Globodera pallid (Urvil et al., 1995, as cited by
Gutierrez-Campos et al. 1999). In the current work, the authors transform
tobacco plants with the oryzacystatin gene and then challenge the transgenic
plants with tobacco etch virus (TEV) and potato virus Y (PVY). Both
viruses are members of the potyvirus group.
The method is as follows:
The following steps were used to construct the chimeric gene and transform it into plants:
Obtain materials, including plant material, bacterial strains and plasmids. The plant materials are kanamycin-resistant tobacco transgenic lines. Tobacco plants are grown in kanamycin-containing medium; only resistant plants survive. These are then self-fertilized to obtain the R1 progeny. The bacterial strain is obtained by subcloning the cDNA from oryzacystatin I into the vector pKYLX80 downstream of the cauliflower mosaic virus 35S promoter.


Figure 1. These are images of real plasmids.
The images can be found at http://www.thermomicro.com/apps/lifesci/lifesci17.htm
Transformation. The plasmid pKLYX80:oryzacystatin is transformed into tobacco leaf disks (obtained from the kanamycin-resistant progeny described above). To see a basic example of a plasmid insertion into bacteria cells, click here.
Now the leaf disks contain cells that are kanamycin-resistant and have the oryzacystatin I gene inside the vector. To verify the presence of the oryzacystatin gene in the transformed cells, the following steps are performed:
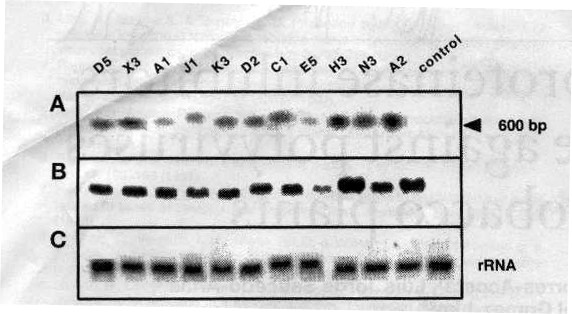
Southern and northern blots. Leaf total DNA of 11 independent transgenic lines was digested with BamHI and electrophoresed. Hybridization was carried out with radioactively-labeled oryzacystatin cDNA (see how hybridization works by clicking here). Leaf total RNA was also subjected to electrophoresis and hybridization; the label again was oryzacystatin cDNA.
The authors found that in the Southern blot, DNA of the transformed cells (Figure 2A, all strains except for control) contained a band that was not present in untransformed cells (control). This is evidence of the transgene. The northern blot was used to analyze the steady-state level of the oryzacystatin message. This message was absent in control plants (Fig. 2B). As a control for equal loading of RNA per lane, the authors hybridized the cells with an rRNA probe (Fig. 2C). This probe did not select for oryzacystatin, and thus the control lane was loaded correctly, as was every other lane on the gel.
Now the transgenic lines must be tested to see if they express inhibition against a cysteine protease. The authors chose papain as their protease.
Assay for papain inhibition. Prepare crude leaf extracts of the transgenic plants and add them to purified papain. The inhibitory activities of the crude extracts are measured by determining the remaining papain proteolytic activity for each line.
In this case, the transgenic lines did indeed inhibit papain. In Figure 2B, the lines showing the highest (H3) and lowest (E5) levels of oryzacystatin mRNA corresponded to those expressing the highest and lowest levels of papain inhibition (data not shown). There is one final step to perform, a real-world application of the research.
Inoculation of plants with viruses. Inoculate plants of the R1 progeny of the transgenic lines with sap from tobacco plants infected with TEV or PVY. Then look for symptoms of the viral infections.
The method works. The plants in this study were resistant to both TEV and PVY. In the past, PDR had been used effectively to increase plant resistance to viruses. Now, this new method can be used to overcome some of the limitations of PDR. Resistance to a variety of different potyviruses can be acquired through one method, a clear benefit over the single-virus resistance offered by PDR.
1.) Gutierrez-Campos, Torres-Acosta, Saucedo-Arias, and Gomez-Lin.
"The use of Cysteine protease inhibitors to engineer
resistance against potyviruses in transgenic tobacco
plants,"
Nature Biotechnology. 17, 1223-1226. 1999.
2.) Lomonossoff, G. P., "Pathogen derived resistance to plant viruses," Annual Review of Phytopathology. 33, 323-343. 1995.
2.) White, Brian. 1995. MIT "Southerns, Northerns, Westerns, & Cloning: 'Molecular Searching Techniques.'" <http://esg-www.mit.edu:8001/bio/rdna/rdna.html > Accessed 2000 February 20.
3.) "Supercoiled DNA." Thermomicroscopes. < http://www.thermomicro.com/apps/lifesci/lifesci17.htm> Accessed 2000 February 20.
4.) Campbell, N. A. Biology, 4th ed. Menlo Park, CA: Benjamin/Cummings
Publishing Company, Inc., 1996.
p. 372-374.
back to Molecular
Biology Home Page
email me at erferguson@davidson.edu