This web page was produced as an assignment for an undergraduate course at Davidson College.
Sex Differences in the Gut Microbiome

From the August, 18 2012 cover of "The Economist"
The following is a review of the article "Sex Differences in the Gut Microbiome Drive Hormone-Dependent Regulation of Autoimmunity" by Janet Markle et al. A full citation and link to the article can be found at the bottom of this webpage. All quotations, facts and data presented in this review are drawn from this article unless otherwise cited.
A Brief Summary...
Autoimmune disorders, such as type 1 diabetes, have been shown to be more prevalent in women. Markle et al. look at this discrepancy and begin to untangle the relationship between such sex biases, microbiomes, and hormone levels. At the center of the paper lies the observation that nonobese diabetic (NOD) mice kept in GF (germ free) conditions do not exhibit the autoimmune disorder sex bias towards females. Conversely, mice kept in SPF (specific pathogen free) conditions do exhibit the sex bias towards female development of T1D. Thus, it seems that microbes are somehow involved in the different susceptibilities of the sexes to autoimmune diseases.
To further analyze this relationship, Markle et al. galvage female mice with either female or male gut microbiomes. The analysis of these mice show that female rats gavaged with male microbiomes exhibit T1D less frequently than untreated females and females gavaged with female microbiomes. Females gavaged with male microbiomes are also shown to have lower instances of T1D precursor phenotypes, such as insilitus (inflamed islet cells) and insulin-specific autoantibodies (Aabs). These results suggest that differences in microbiomes are at play in the sex-dependent susceptibilities to T1D and other autoimmune disorders.
Additionally, Markle et al.’s data indicate that the transplanted male microbiomes affect the recipient female’s testosterone levels. Females gavaged with male microbiomes exhibit higher testosterone levels than control females and those that receive female microbiomes. When females gavaged with male microbiomes are treated with a testosterone inhibitor, their increased protection from T1D and its precursor phenotypes decrease.
Evaluation
This paper does a lot. This five-page paper represents the conglomeration of a dozen different experiments. As a result of cramming so much information and data into a single paper, the authors cannot possibly address every interesting aspect of a problem. Rather, they must pick the story they are trying to tell and follow it throughout their different datasets.
Having so much information present in one paper means that at times, the authors seem to ignore interesting results that seem to apply to their problem. For example, the differences between female testosterone levels in SPF and GF environments in Figure 1 or what sometimes seems to be increased susceptibility to T1D and precursor phenotypes when female mice receive female microbiomes (Figure 1B, C). This can sometimes be frustrating, especially when the authors do not acknowledge that they cannot address that aspect.
That said, the authors do an excellent job of creating a narrative within their paper. They spell out the reasoning behind each experiment and relate their results back to those presented previously to create a singular strain of thought. The logic underlying the experiments and especially the construction of the paper is quite elegant and well thought out. Such an engaging narrative ensures that the points that they intend to make come across strong.
Perhaps this papers greatest weakness was a lack of attention to what data was shown. In Figure 1, changes in the microbiome are shown for control M vs. control F, male donor/female recipient vs. control F, and male donor/female recipient vs. female donor/female recipient, but not male donor/female recipient vs. control male. Since much of the authors argument rests on the fact that the microbiome created by gavaging a female mouse with a male microbiome is unique from both female and male microbiomes, this would have been a logical comparison to include. Not showing this data weakens the strength of their paper.
Overall, I thought this was a really strong paper. I enjoyed reading it; I found their logic both engaging and easy to follow along. The only complaints I had dealt with figure details, which are addressed in the individual figure analyses below, and the above control concern.
Figures
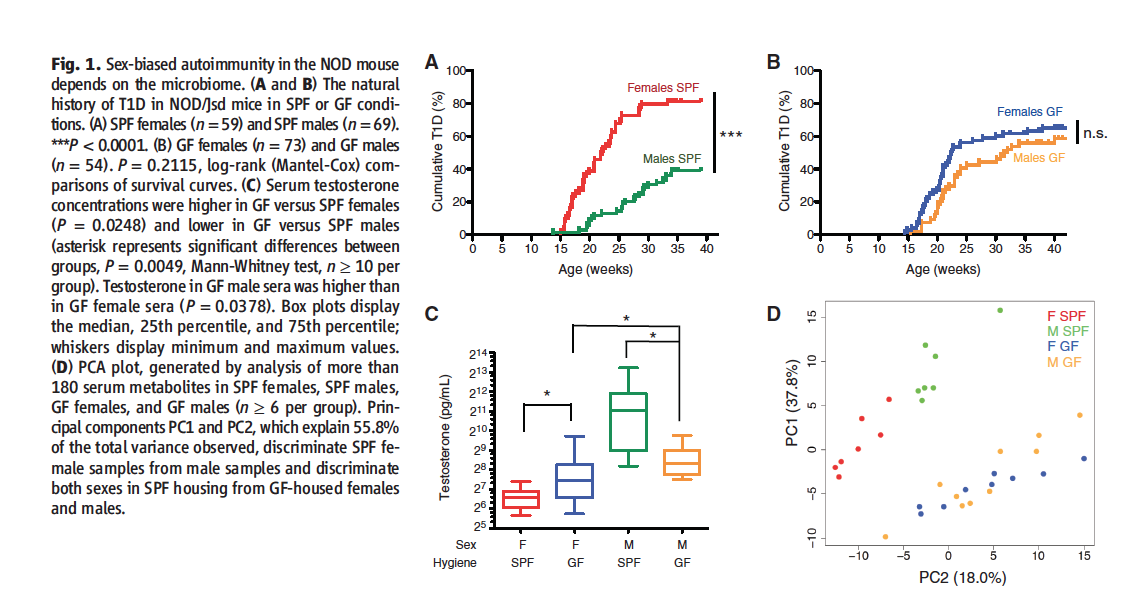
Figure 1 summarizes the data researchers used to justify focusing on gut microbiomes as potential factors at play in sex differences in the regulation of autoimmunity.
Figures 1A and B plot the age of the mice against the cumulative incidence of T1D for both males and females kept in SPF and GF conditions. In the SPF condition, significantly more female mice developed T1D than male mice (~80% vs. ~40%). In GF conditions, male incidence of T1D rose to match the female. In GF conditions, there is no significant difference between the male and females, both appear to be ~%60. However, the authors do not indicate if the female incidence of T1D is significantly different in GF vs. SPF conditions, therefore whether ~%60 is significantly different than ~%80.
Figure 1C plots the testosterone concentrations in male/female mice kept in SPF or GF conditions. Male SPF mice testosterone levels are significantly higher female SPF mice and both sexes of GF mice. Male GF mice testosterone levels are significantly higher than female GF mice, which is to be expected. Interestingly, however, females kept in GF conditions had significantly higher testosterone levels than females kept in SPF conditions.
Figure 1D depicts principle component analysis of serum metabolites from all four conditions. PC1 and PC2 contain a subset of glycerophosopholipid and sphingolipid metabolites that account for most of the variability between the groups. When the two are plotted against each other, SPF males and females cluster into distinctly separate groups. Such clustering does not occur between GF males and females.
Take Home Message: Male NOD mice kept in SPF environments develop T1D less frequently than their female counterparts. This sex-based difference disappears when the mice are kept in GF environments. Analysis for testosterone reveals higher levels of the sex horomone in male mice kept in SPF environments as opposed to GF conditions. Additionally, PCA analysis of metabolites reveals distinct male/female clustering in SPF but not GF conditions. Taken together, these results indicate that a sex-based difference in the the development of T1D in NOD mice is as a result of some sort of microbe presence, potentially affecting testosterone levels.
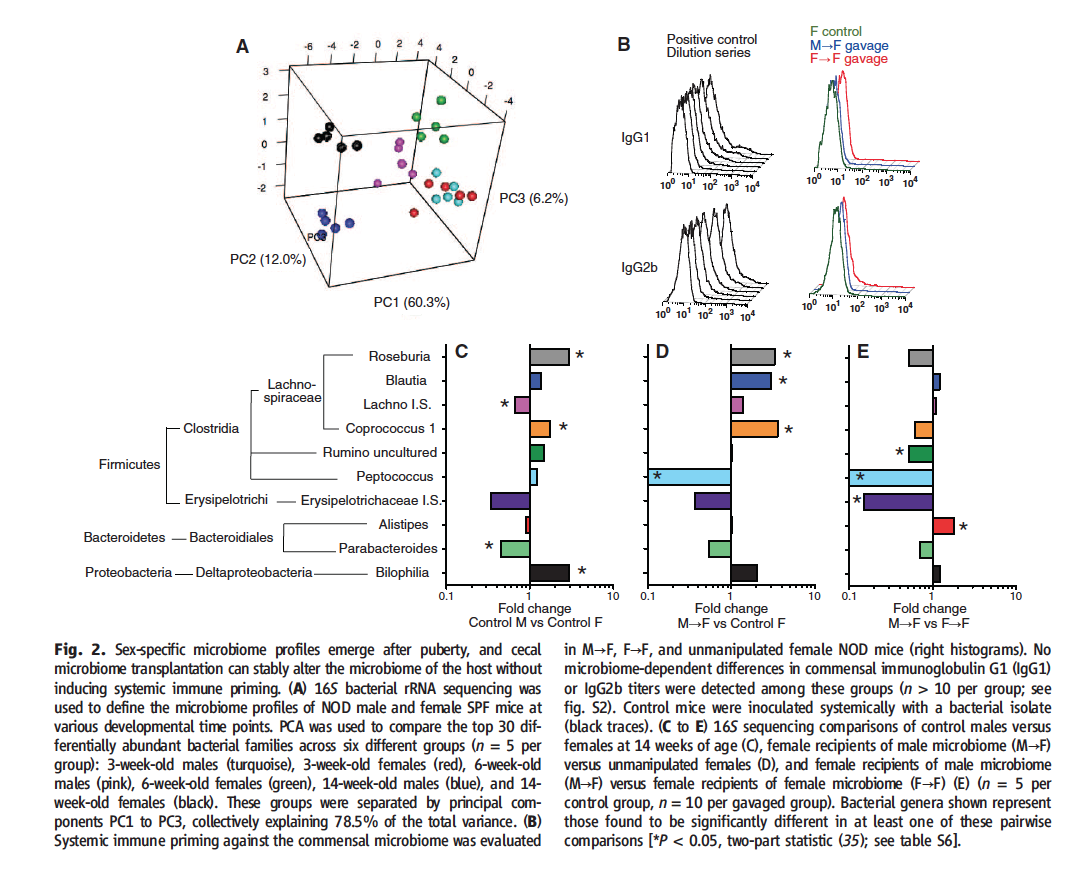
Figure 2 represents the data researchers used to claim that by gavaging young female mice with the cecal contents of adult male mice, they created a third gut microbiome, distinct from those of both adult male and female mice.
Figure 2A depicts the PCA done on bacterial rRNA obtained from the cecal contents of male and female mice at weaning, puberty, and adulthood. Sex-specific clusters are evident at puberty and even more so at adulthood, but not at weaning. These results suggest that sexual maturation affect the cecal microbiome.
In Figure 2B, researchers show that their methods did not result in antibodies specific for the bacteria weanlings were exposed to. Previous studies have shown that microbe exposure can result in some sort of protection from the NOD mice development of T1D. In this figure, researchers show that since the mice didn’t develop antibodies to the bacteria they were exposed to, this phenomena was not at play.
Figure 2C-E Researchers transplanted male or female donor cecal contents into host weaning female mice. Analysis occured at 14 weeks for experimental and control condtions. The figure depicts fold changes for control males vs. control females (C), male donor/female hosts vs. control females (D), and male donor/female host vs. female donor/female host (E). The phylogentic tree displayed to the left represents some of the bacterial genera present that were found to be significantly different in at least one of the comparisons researchers were making. It is interesting that a large number of the bacteria levels found to be significantly different were in the same genera (Firmicutes). However, I still find this part of the figure to be superfluous and not necessary to the main point of the figure. Researchers use the data presented in this part of figure two to support their assertion that by transplanting male cecal contents into a female host, they have created a unique third microbiome state. However, they do not show a comparison of male donor/female host to control males, which would be helpful in evaluating the researcher’s claim. If the issue at play was valuable page space, I would have rather seen this comparison than the space-intensive phylogentic tree. Later analysis showed these changes were not present at age 34 weeks.
Take Home Message: The cecal microbiomes of male/female mice at weaning are indistinguishable from each other. However, distinct sex-based clusters become evident as sexual maturation occurs. Researchers gavaged young female mice with the cecal contents of adult NOD male or female mice, essentially transplanting a microbiome. Researchers then compared the new, manipulated microbiome to controls to show that they created a unique, new microbiome by performing this procedure.
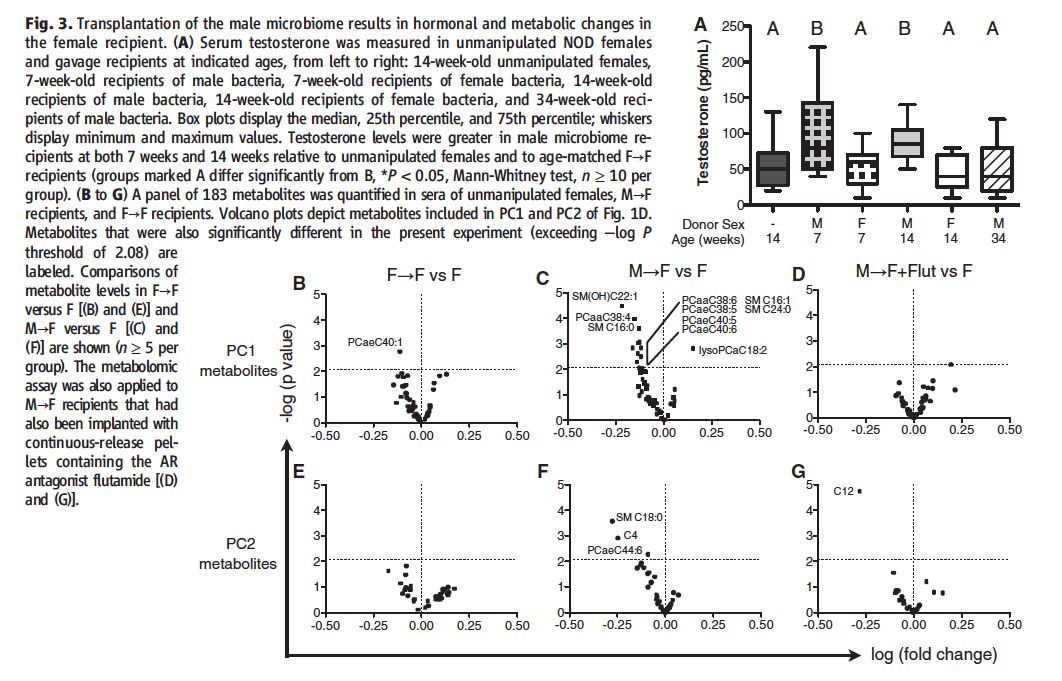
Figure 3 examines the effects of altering mouse microbiomes on testosterone and metabolites.
Figure 3A Researchers tested testosterone levels females who were gavaged with either male or female cecal contents. Results show that female host mice who received cecal contents from a male mouse showed significantly increased levels of testosterone at 7 and 14 weeks of age, but not 34 weeks of age, when compared to 14 week old unmanipulated control female mice and to mice who received cecal contents from a female donor.
Figure 3B-G Researchers performed PCA on the metabolomes of 14 week old unmanipulated controls, female donor/female recipient, male donor/female recipient, and male donor/female recipients treated with flutamide, a testosterone inhibitor. The flutamide group was added to ascertain whether or not the increased testosterone levels observed in Figure 1 were at play. Since flutamide is a testosterone inhibitor, it essentially negates testosterone’s effect on the system. The graphs shown plot the PC metabolite levels against fold change for the control group versus all three experimental conditions. The results show that when the microbiome comes from a male donor, a unique metabolome is established when compared to the untreated controls. This metabolome is different from the metabolome created when a female donor micrbiome is utilized. When the male donor group is treated with testosterone, the metabolome becomes more similar to that of the female donor group than the male donor group untreated with flutamide. This suggests that testosterone is a major player in altering the metabolic activities of the mice.
Take Home Message: Female NOD mice who are gavaged with male microbiomes exhibit altered testosterone levels and metabolomes. When a testosterone inhibitor is administered, the metabolome of the mice is altered less drastically. These results imply that testosterone acts as some sort of metabolic regulator.
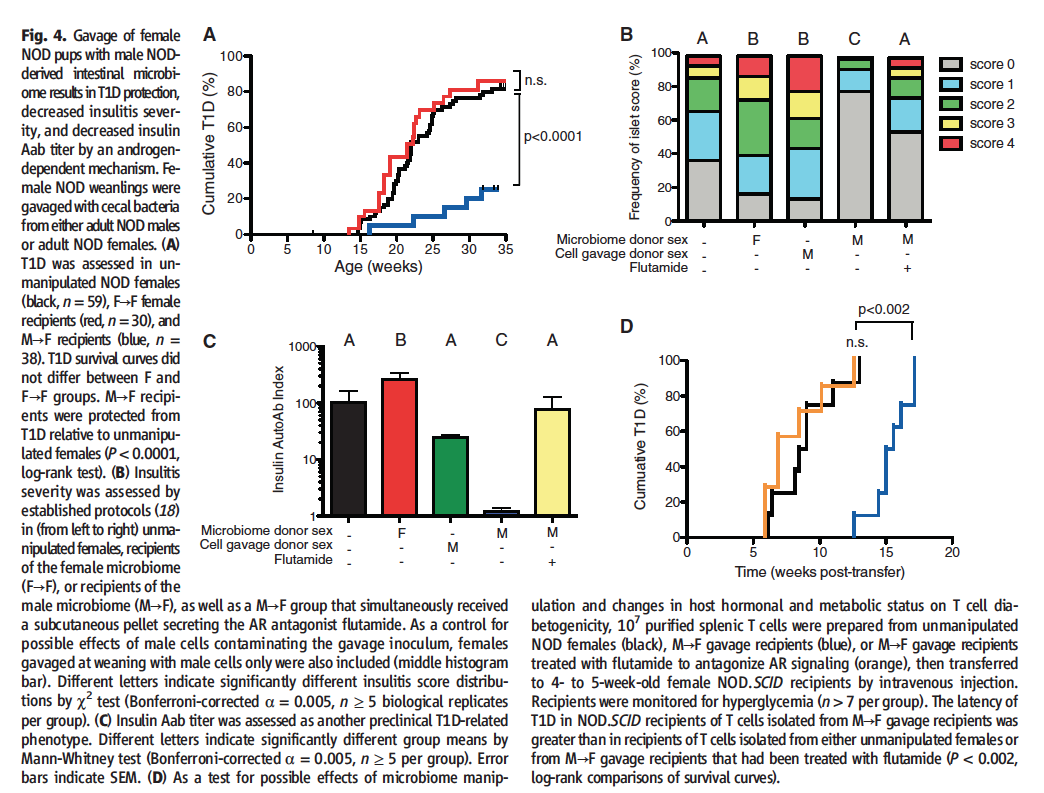
Figure 4 includes data that researchers used to support their claim that gut microbiomes and testosterone are at play in the regulation of autoimmunity.
Figure 4A shows the incidence of T1D in females gavaged with male microbiomes, females gavaged with female microbiomes, and untreated females. Analysis reveals a decreased incidence of T1D when the microbiome donor is male.
Figure 4B shows the prevalence and severity of insulitis, a precursor to diabetes.The color coding denotes the severity of the insulitis. The legend to the scale is not given in the paper; the reader is referred to online supplementary material. However, I could not find the legend to the score scale there either. From the authors interpretation of the figure in the text, I have inferred that the scale goes from normal (0) to less severe( (1) to most severe (4). Perhaps this scale is well known to many scientists, but in my opinion it would have been simpler to just use the terms most severe, less severe, normal etc. in the figure legend. Using this interpretation of the scale, the more color (scores of 1-4) a bar has, as opposed to gray (a score of 0), the higher the incidence of insulitis. The amount of each color within the bar allows for interpretation of the severity of the islet of the inflammation. Mice that received male microbiomes exhibited decreased incidence of islet inflammation, results congruent with the decreased incidence of T1D displayed in Figure 4A. When female mice that have received male donor microbiomes are administered flutamide, a testosterone agonist, this protection is lost, results that are also congruent with 4A. In order to ensure that the observed reduced susceptibility to T1D was not an immune response, as discussed in Figure 1, researchers gavaged females with sterile male cells only. These sterile cells had no bacteria, researchers were trying to see if the female immune system was recognizing the male cells as foreign, mounting an immune response to their presence and thus resulting in the observed decreased occurrence of T1D. However, as the data displayed here shows, that was not the case; the group gavaged with sterile male cells exhibited no increased protection from islet inflammation, indicating that no antibodies are being produced. In fact, both this group and the group that received microbiomes from a female donor seem to exhibit more islet inflammation than the control group, a phenomenon the authors do not discuss.
Figure 4C Insulin specific Aab (autoimmune antibodies) are another precursor to diabetes in NOD mice and humans. Here, researchers quantified the amount of insulin-specific Aap, and found that female mice that received male donor microbiomes exhibited significantly lower levels of insulin Aabs than all other groups. The control group, the group gavaged with sterile male cells, and the group that received both male donor microbiomes and flutamide exhibited no significant difference from each other. Those that received female donor microbiomes exhibited a significantly higher incidence of insulin Aabs, a phenomenon the authors do not address.
Figure 4D Researchers were concerned about unintentional effects of the microbiome manipulations specifically on T cells. Thus, researchers took T cells from unmanipulated controls and male donor/female recipient (both with and without flutamide) conditions and administered them to NOD.SCID (non-obese diabetic, severe combined immune-deficient) mice. The incidence of T1D was then recorded. The unmanipulated control and flutamide treated male donor conditions transferred T1D to their recipient hosts along equivalent timelines. However, it took the T-cells isolated from male donor/female recipient mice significantly longer to transfer T1D. This once again implicates testosterone as a major player in autoimmunity.
Take Home Message: Female mice that gavaged with male microbiomes exhibit a lower incidence of T1D and exhibit prediabetic phenotypes (such as insulitis and insulin Aabs) less frequently than the control group and male donor/female recipient mice treated with flutamite, a testosterone inhibitor. Additionally, T-cells from the control group and experimental group treated with flutamite transfer T1D to recipient NOD.SCID mice faster than those isolated from the male donor/female recipient group. This indicates that the microbiomes of the mice are somehow affecting testosterone levels, which seem to regulate autoimmunity.
References
Genomics Page
Biology Home Page
© Copyright 2014 Department of Biology, Davidson College, Davidson, NC 28035