This web page was produced as an assignment for an undergraduate course at Davidson College.
Induced Pluripotent Stem Cells

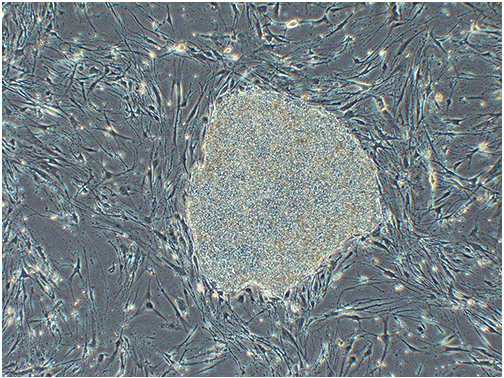
iPSC image from the RIKEN Center
Permission Granted
Human embryonic stem cells (hESCs) were grown in a lab for the first time in 1998. Since then, stem cells have represented limitless possibilities for potential therapies and research to the scientific community. One of the most promising types of therapies involved using human embryonic stem cells to treat degenerative diseases such as Parkinson's or Alzheimers. Others hoped to eventually grow whole organs from hESCs as an alternative to traditional organ donor sources. However, there are problems associated with hESCs. For some, the fact that hESCs must be taken from days old human embryos represents an ethical dilemma that outweighs the potential benefits of hESC research. Additionally, the threat of immune rejection is a very real obstacle to the pathway of many potential therapies involving hESCs. The development of induced pluripotent stem cells (iPSCs) represents the most recent step fowards in stem cell research (Stem Cell Basics). Researchers first reported iPSCs derived from mouse in 2006(Takahashi and Yamanaka 2006). iPSCs from humans were first reported in 2007 (Takahashi et al 2007). Induced pluripotent stem cells are on the breaking edge of stem cell research and may hold the key to integrating stem cell therapies into modern medicine.
What are induced pluripotent stem cells?
Induced pluripotent stem cells are adult, differentiated cells that scientist reprogrammed to behave in a similar manner to hESCs. Pluripotent cells have the ability to differentiate into any kind of somatic cell. iPSCs can undergo an unlimited number of cell divisions without losing their pluripotency (Romano et al 148).
How does "induced" work?
In order to create pluripotent cells from differentiated somatic cells, factors must be introduced into the cell that cause it to turn on/off the appropriate genes for pluripotency. These include various transcription factors normally expressed in embryonic cells. Currently, most researchers use viruses to introduce these factors into somatic cells. Once introduced, the factors become incorporated into the host cell’s genome and are expressed along with the host’s own genes (Romano et al 148).
What can we do with these cells?
One of the most exciting applications of iPSC research is organ transplants. The number of patients who would benefit from organ transplant far exceeds the number of organs available. iPSCs hold the potential to allow for the development of replacements for defective or degenerative tissues from a patient’s own somatic cells. Not only would this increase the number of organs available for transplant, but also could also limit the number of organ rejections. Currently, transplant patients must undergo rounds of immunosuppression therapies to limit their immune system’s response to foreign tissue. If organs or tissue are developed from a patient’s own cells, the patient’s immune system may not identify the new, healthy tissue as foreign and not attack it (Stem Cell Basics).
Japanese researchers are currently conducting the first iPSC therapy trial in humans. They are using retinal cells derived from iPSCs to treat age related macular degeneration. Current treatment for the disorder involves halting or significantly slowing the degeneration, but not repairing the damage done. In this trial, researchers will first remove the degenerative retinal tissue from one eye and then grafting iPSC derived retinal cells to the damaged area of the same retina. The idea is that the retina will functionally integrate these healthy cells, repairing lost function. The other eye is left untreated as a control (Riken 2013). This represents the first of many to come exciting forays into iPSCs as treatment for many diseases.
iPSCs hold potential not only as a therapeutic tool, but also offer unique research opportunities. Since by definition, iPSCs can undergo indefinite rounds of cell division, they could shed light on the intricacies of the cell cycle. Abnormalities in the cell cycle and differentiation play a role in many human diseases and defects. A more complete understanding of the genetic and molecular regulations of the cell cycle could result in new therapies and approaches for treatment of some of these diseases (Potential Uses, 2009). Some researchers have manipulated iPSCs to create models of human diseases for which there are no or limited animal models. These include many cardiovascular diseases, neurodegenerative diseases, and psychiatric disorders (Romano et al 150). iPSCs may also be used to screen newly developed drug therapies, creating another way to test a drug’s effectiveness and safety without risking human health (Romano et al 151).
What hurdles does iPSC research have to overcome?
One potential limiting factor to iPSCs is the DNA abnormalities inherent in being derived from human somatic cells. These cells have probably already undergone rounds of cell division and been exposed to environmental toxins, potentially damaging their DNA. Additionally, there are complications with the current viral method of the introduction of transcription factors. This method often disrupts the cell’s genome in such a way that result in malignancy. Viruses are also difficult to control and regulate. Research is underway to develop non-viral methods of transcription factor introduction (Romano et al 149).
See NIH.gov for more information on iPSCs
References
Riken. "Pilot clinical study into iPS cell therapy for eye disease starts in Japan." July 30, 2013. Available here.
Roman, Gaetano et al."A Commentary of iPS Cells: Potential Applications in Autologous Transplantation, Study of ILlnesses and Drug Screening." Journal of Cellular Physiology 229(2); 148-152. 2013.
Stem Cell Basics. In Stem Cell Information [World Wide Website]. Bethesda, MD: National Institutes of HEalth, US Department of Health and Human Services, 2009. Available here.
Stem Cells and Diseases. In Stem Cell Information [World Wide Web site]. Bethesda, MD: National Institutes of Health, U.S. Department of Health and Human Services, 2012 Available here.
Takahashi, K. Yamanaka, S. "Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors." Cell, 126(4). 2006.
Takahashi, K et al. "Induction of pluripotent stem cells from adult human fibroblast cultures by defined factors." Cell, 131(5). 2007.
Assignment #2:You get to choose (due Feb. 28, 2 pm)
Assignment #3: You get to choose (due Apr. 25, 2 pm)
Genomics Page
Biology Home Page
© Copyright 2014 Department of Biology, Davidson College, Davidson, NC 28035