The main point of this paper was to
determine if admixture of Neandertal alleles in populations of
Eurasian descent could explain variations in disease and disease risk
related phenotypes. They
were able to perform this study using publically available data from
the Electronic Medical Records and Genomics (eMERGE) Network, which
links genetic data from nine locations in the US to electronic health
records (ERH). Using this network, they were able to analyze 28,416
individuals (randomly split into discovery and replication cohorts).
After generating a constrained list of
SNPs that were most likely to derive from Neandertal alleles, the
researchers first performed a genome-wide complex trait analysis
(GCTA) on 1495 common Neandertal SNPs. They found that Neandertal SNPs
explained a significant amount of risk in three traits: depression,
myocardial infarction, and corns and callosities. Another nine traits
were nominally, but not statistically, significant. Eight of these
twelve traits were replicated in the second cohort, and three remained
significant when compared to non-Neandertal SNPs.
The GCTA analysis allowed the
researchers to observe how phenotypes correlate with a large number of
SNPs, but they also wanted to find if single loci affected human
phenotypes. To do this, they had to perform a phenome-wide association
study (PheWAS). This found four SNPs which explained significant risks
for various phenotypes:
o
SNP
in a intron of SELP, which was significantly associated with a
hypercoagulable state
o
SNP
in an intron of SLC35F3, a thiamine transporter, which could
exacerbate the effects of malnutrition
o
SNP
upstream
of STIM1, an intracellular calcium transporter, associated with loss
of bladder control
o
SNP
in an itron of SLC6A11, a neurotransmitter transporter, associated
with nicotine addiction
They also demonstrated that SNPs do not affect all phenotypes equally.
Neandertal SNPs are significantly enriched for neurologic and
psychiatric phenotypes while being under represented in digestive
phenotypes.
Overall, the researchers accomplished what they set out to do,
correlating medically relevant phenotypes to Neandertal derived SNPs.
The researchers note that there are some limitations to their analysis
because they can only look at phenotypes present in health records,
meaning that any traits related to appearance or behavior remain
largely uncorrelated with Neandertal derived SNPs.
I chose this paper because I
thought that the implications of “borrowed” Neandertal alleles on
modern populations could have practical application in evolutionary
biology, while the methods could lead to further genetic information
about why different populations are more susceptible to various
diseases. The paper did a good job of convincing me that their methods
reduced the possibility of false discovery and we reproducible.
However, there were certain aspects were I found the paper to be
lacking. First, I would have liked the paper to go into more detail
about what “Eurasian descent” meant. Are the effects still significant
in Southeast Asian and Oceanic populations? Could they have used
populations of sub-Saharan descent as examples of populations that
don’t have the disease causing alleles and are also less likely to
develop the specific phenotypes addressed? These questions, for me,
remain unanswered.
Additionally, I thought that some of the statistics used to verify that
their findings were significant were difficult to understand. The S*
statistic and BLUP specifically come to mind. S* is mentioned once in
the paper when they are trying to confidently identify SNPs that come
from Neandertal haplotypes. S* is not described in the paper, nor is
it a common statistic. S* was only able to be identified by reading
the reference sited in the paper. The S* statistic turns out to be a
measure of linkage disequilibrium “to identify which SNPs are the most
likely to have mutated in a putative archaic population” (Plagnol
& Wall, 2006). Best
linear unbiased prediction, or BLUP, was used to estimate the effects
of individual SNPs, but again, it is unclear to me how this works.
Lastly, I thought that the authors could have been a little clearer early
on in the paper that the ERH were integrated with genomic data via
eMERGE networks. I began reading the paper thinking that these were
two different sources, and only resolved the distinction upon closer
reading.

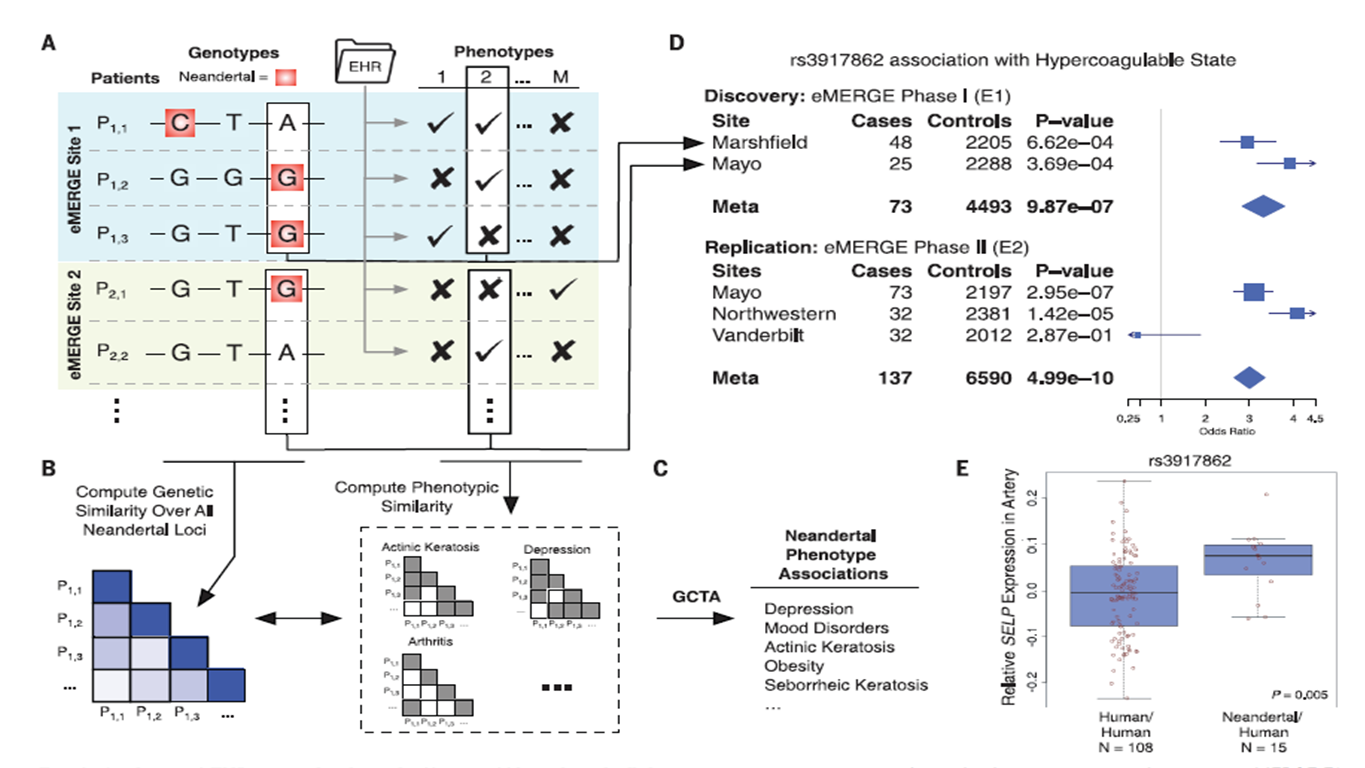
Figure
1. This figure gives a broad overview of the methods they
used in this study. (A)First, they took genomic data and isolated
Neandertal SNPs. Then, they correlated SNP with phenotypes with EHR
data using eMERGE. They then used this data to derive phenotypic
similarity for complex traits using GCTA (B & C), and phenotypes
that could be derived from single SNPs using PheWAS (D & E). The
point of this figure is not necessarily to show the significance of
any trait, but more to draw to the reader’s attention that their
methods involve multiple analyses and produce significant results.
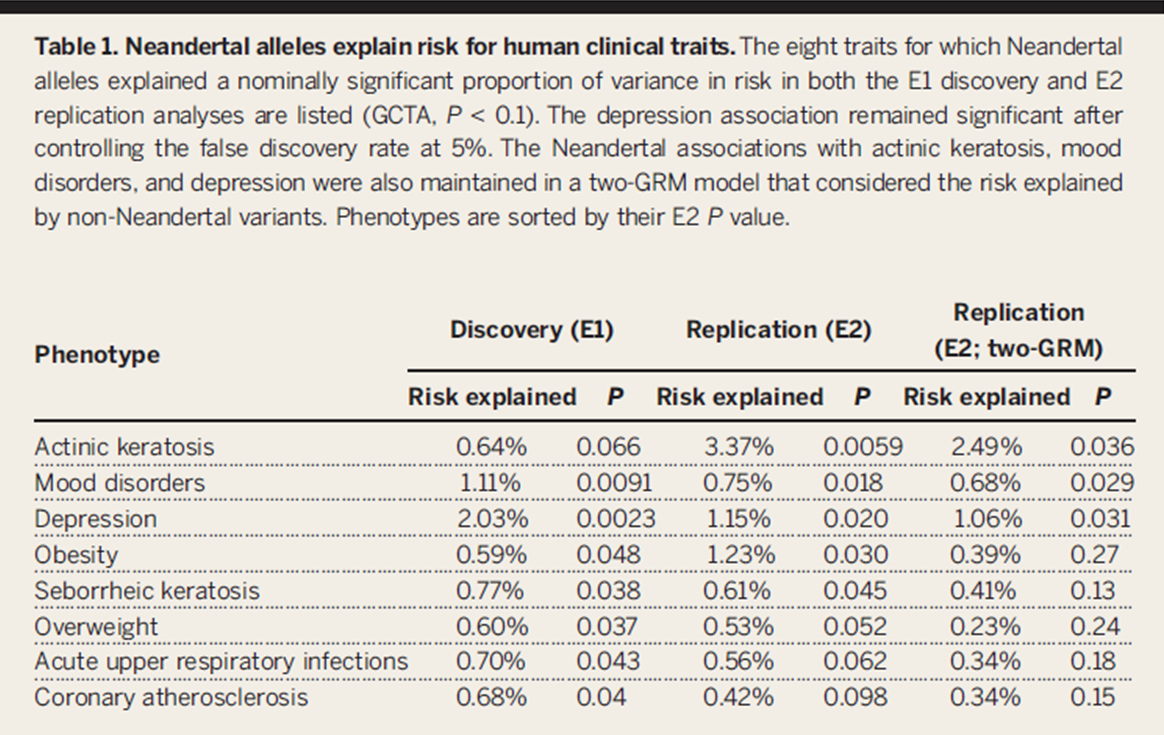
Table
1. This figure highlights the eight traits found by their
GCTA method to explain at least a nominally significant (p < 0.1)
percent of the risk in both the discovery and replication cohorts.
They also performed a genetic relationship matrix (GRM) test to
compare the percent of the risk explained by Neandertal SNPs to that
explained by non-Neandertal SNPs. After this, 3 traits still remained
statistically significant. This shows that they are able to replicate
their results and still produce significant results under strict
criteria.
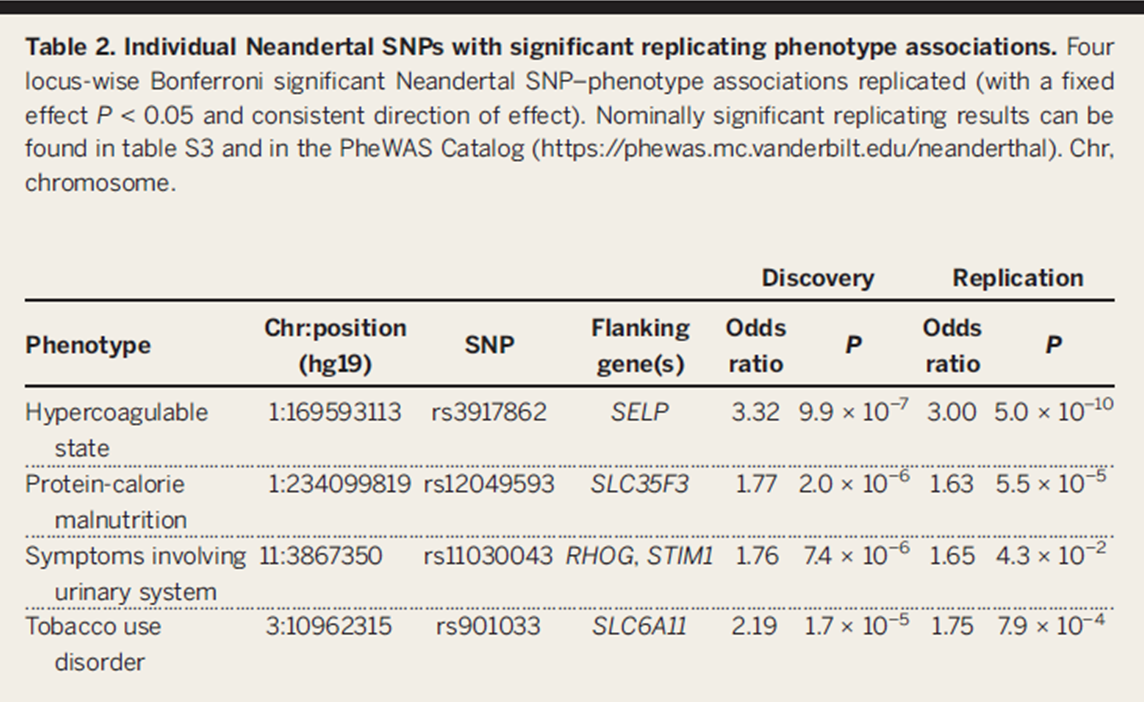
Table 2. This figure shows the 4 phenotypes found by their PheWAS method which have significant correlation with Neandertal SNPs. In this method, they used a Bonferroni model of significance, which allowed them to determine significance in their results while testing many hypotheses simultaneously. Again, they show that they are able to replicate significant results in each of their independent cohorts.
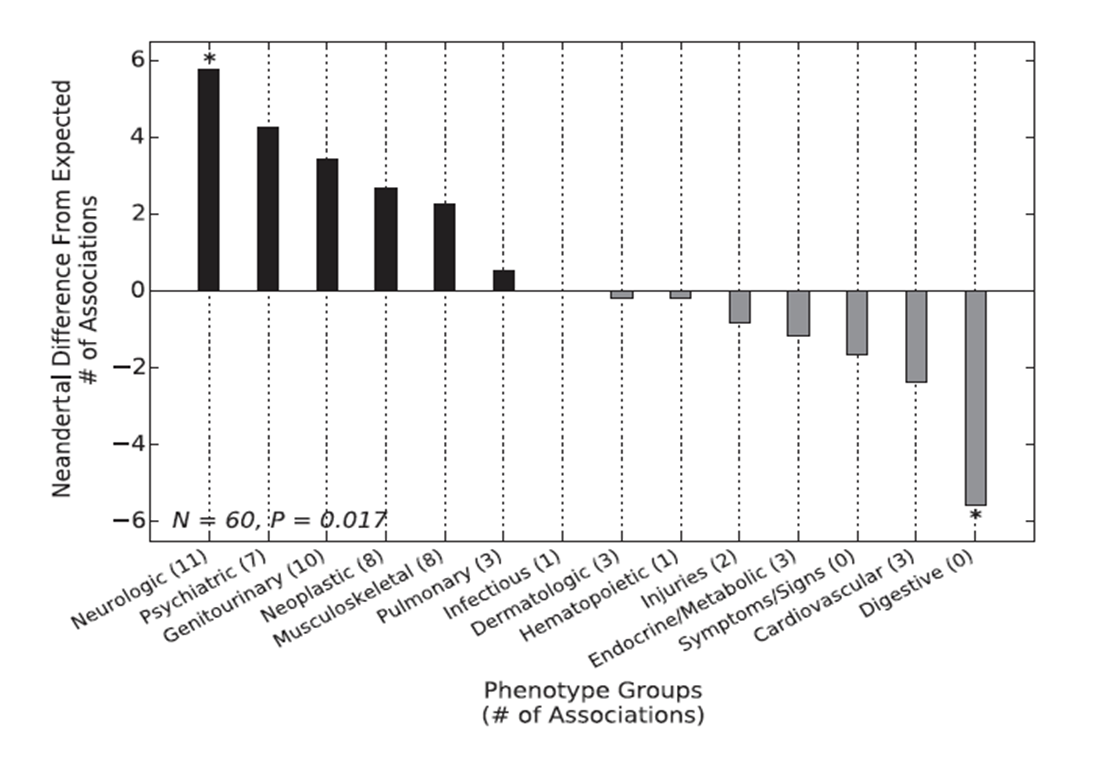
Figure
2. In this figure, the researchers performed another PheWAS
study to test for enrichment of Neandetal SNPs on specific types of
phenotypes. To do this, they compared phenotype association of a set
of Neandertal SNPs to 5 different sets of non-Neandertal SNPs that
occurred at similar frequencies as the Neandertal SNPs. This showed
that Neandertal SNPs are significantly enriched for different groups
of phenotypes than would be expected. Most importantly, notice the
high enrichment of psychiatric phenotypes.
Plagnol, V, Wall, J. 2006. Possible Ancestral Structure in Human Populations. PLOS. http://journals.plos.org/plosgenetics/article?id=10.1371/journal.pgen.0020105. accessed Apr 21, 2016.
Simonti, C, Vernot, B, Bastarache, L, Bottinger, E, Carrell, D, Chisholm, R, Crosslin, D, Hebbring, S, Jarvik, G, Kullo, I, Rongling, L, Pathak, J, Ritchie, M, Roden, D, Verma, S, Tromp, G, Prato, J, Bush, W, Akey, J, & Denny, J. 2016. The phenotypic legacy of admixture between modern humans and Neandertals. Science 351:737-741. http://science.sciencmag.org/contents/351/6274/737.
© Copyright 2016 Department of Biology, Davidson College, Davidson, NC 28035