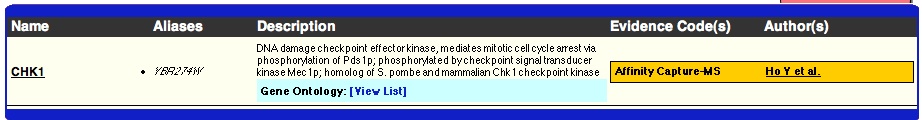
The proteomic information available for YLR152C is sparse. The limited information points towards the protein being found in the nucleus as it was found to interact with a protein kinase, which is located in the nucleus. As the experiment that found this interaction was an affinity capture-MS and not a yeast-two-hybrid, I have more confidence that the protein could exist in the nuclear membrane. As no proteomic data currently refutes the prediction that YLR152C is a membrane protein and Kyte-Doolittle Hydropathy plots predict at least four transmembrane domains (see my favorite yeast gene), more information is needed before we can reject the hypothesis that YLR152C is a membrane protein. The current proteomic data does not seem to support YLR152C working directly in one of the metabolic pathways like glycogen synthesis.
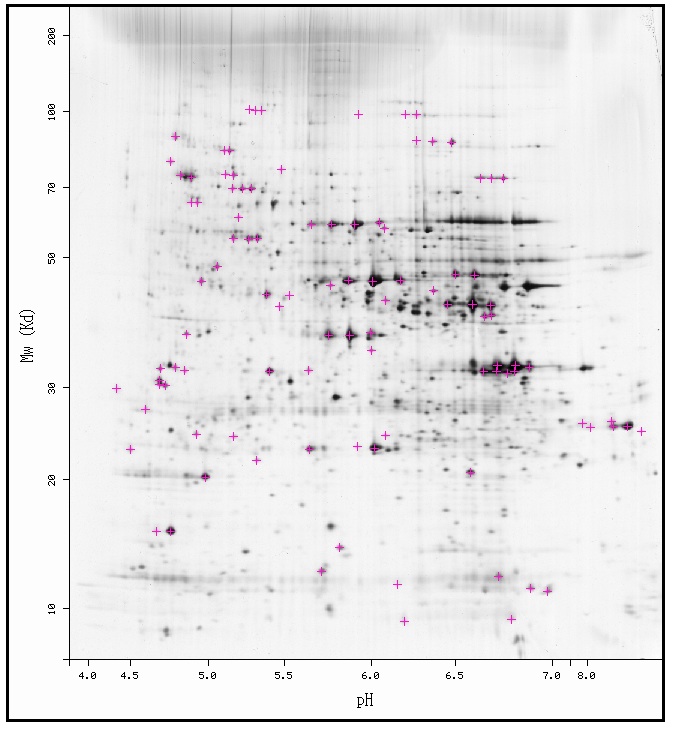
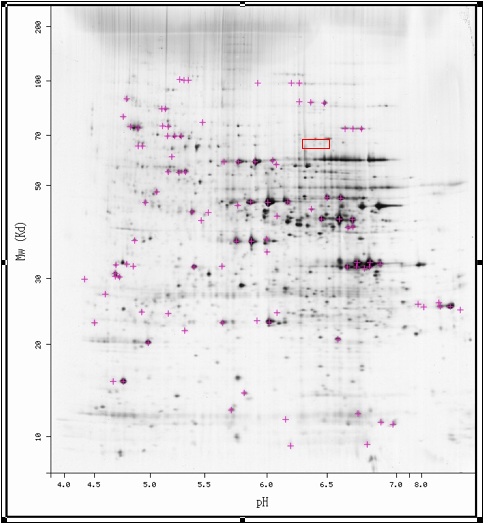
The first experiment that I would suggest is the identification of YLR152C by mass spectrometry. While the 2D-gel had faint spots where YLR152 is predicted to be, YLR152C has yet to be identified and may not actually exist there. If a regular 2D-gel does not contain YLR152C in its predicted area, a second gel should be run using the detergent Triton X-114. Triton X-114 keeps membrane bound proteins suspended in it and allows a 2D-gel to resolve the proteins; thus, YLR152C would probably be resolved using this method if the prediction that it is a membrane protein holds true.
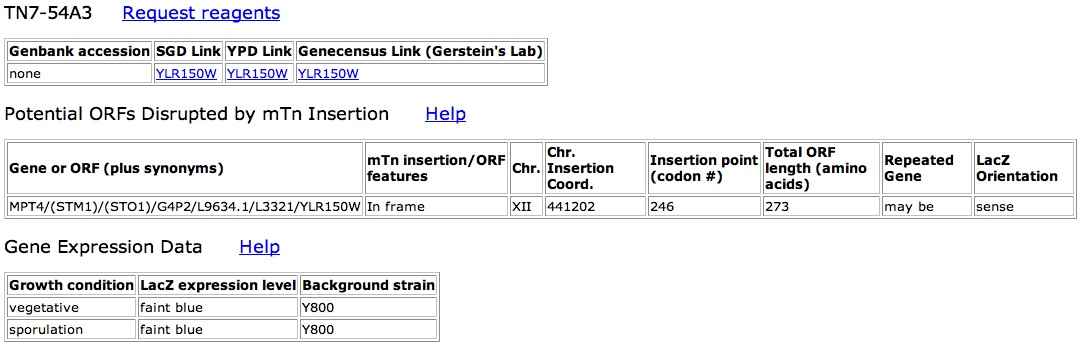
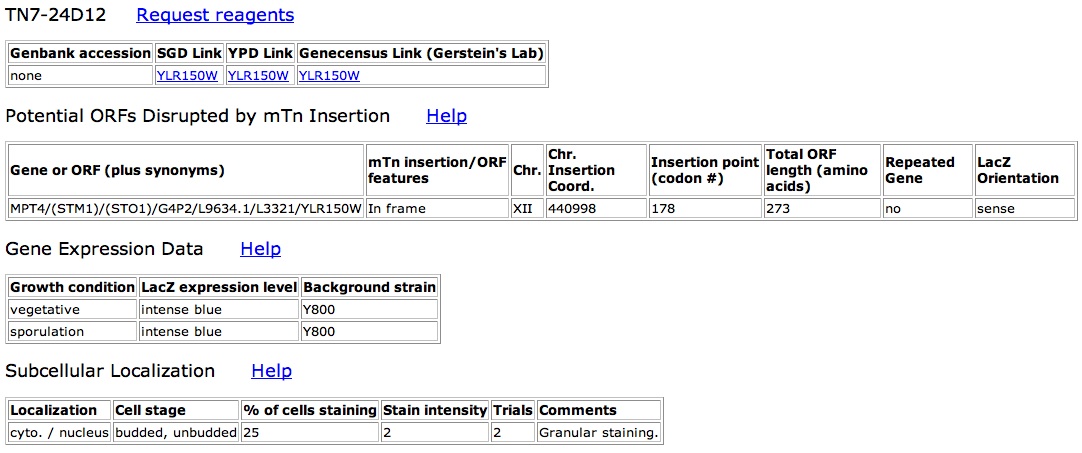
As the only known interaction is with the protein kinase in the nucleus, attempts should be made to find the subcellular localization. I suggest creating mTn disrupted clones. The cells can then be stained to see if there is any localization as has been done with other proteins found in the TRIPLES database. The localization would at least tell if YLR152C does indeed stay in the nucleus and may also be able to discern whether YLR152C exists in the nuclear membrane or in the nucleus at large.
As with STM1, we can gain great knowledge if we perform experiments that check actual protein expression against mRNA expression for the protein. By using the ICAT method with cells grown under conditions that show an induction or repression of mRNA greater than or equal to a threshold value or even under conditions that show any change in expression, we could find what happens to the protein expression for the same conditions. If the two expressions are not the same (RNA shows an induction while the protein remains stable or is repressed and vice versa), we could run further experiments to see if YLR152C is not being activated by phosphorylation or other methods or if it is being activated more efficiently than normal.
While doing the ICAT experiments based on microarrays, we can also check for the change in expression throughout the cell cycle. What a protein does at different times can be very telling about its actual function and purpose; therefore, gathering this information will expand out knowledge of YLR152C.
Finally if the previously suggested experiments strongly refute YLR152C being a membrane protein, researchers should attempt to crystallize YLR152C. As structure has often led to new insights in how and why a protein works, having a structure greatly increase our potential to understand YLR152C. |