*This website was produced as an assignment for an undergratuate course at Davidson College.*
My Favorite Protein:
Insulin
Background
Insulin is a small protein (more specifically a hormone) that lowers blood sugar. Insulin was discovered by Fredrick Banting, Charles Best, J.J.R. Macleod and James Collip (Strakoshc, 2005). At that time, it was know that diabetes was a disease that caused people who were affected to have difficulty controlling their blood sugar levels. Upon their discovery of insulin, a way to battle diabetes was found.

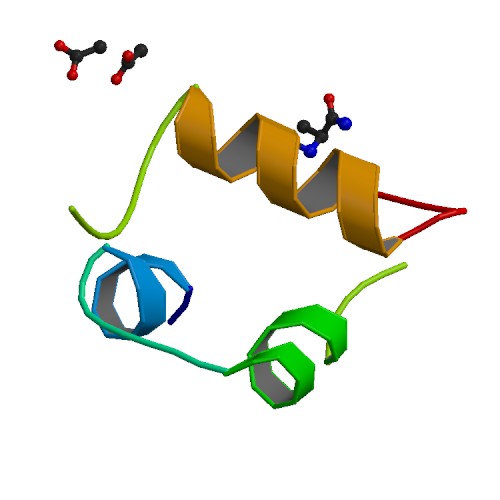
Figure 1.Stucture of hyperactive human insulin analogue courtesy of Protein Data Bank. The bottom strand is the A subunit and the top strand is the B subunit.
Structure of Insulin
The primary structure of insulin is made from two polypeptide chains named subunit A and B. Subunit A consists of 21 amino acids, whereas subunit B consists of 30 amino acids. These chains are connected by two disulfide bridges as seen in figure 2. Insulin also forms quaternary structure by creating diamers using hydrogen bonds and hexamers by bonding with two zinc ions (Bowen, 1999). Insulin The final product of insulin only consists of 51 amino acids so it is quite small compared to other proteins. Insulin's small size allows it to be a ligand for other proteins appropriately named insulin receptors.
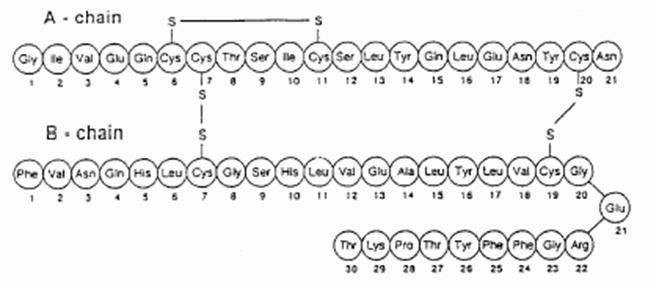
Figure 2. The primary structure of human insulin. As you can see, there are 21 amino acids in subunit A and 30 amino acids in subunit B. Image from:
http://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?id=7043&type=display
Synthesis of Insulin
The insulin gene is only expressed in B-cells in the pancreas (Roy et al., 2003). When this gene is expressed, pre-mRNA called proinsulin is made. This precursor to insulin is only on polypeptide chain and it contains both subunit A and subunit B in it. The A subunit is located at the C-terminus whereas the B subunit is located at the N-terminus The C subunit, that is cut out of the end product, is cut out of the polypeptide by specific proteases (Bowen, 1999).
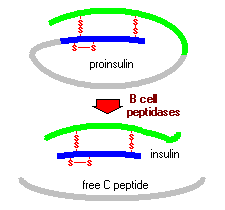
Figure 3. Picture of proinsulin and insulin after it is processed in the endoplasmic reticulum. Image from Bowen, 1999; permission pending.
Function of Insulin
Because of Banting and his colleagues' discovery, insulin was found to help regulate the amount of glucose in the blood. However, insulin serves more functions than just regulating the amount of glucose in one's blood. Insulin also regulates the formation of fatty acids in the liver as well as having catabolic funtion in the oxydization of sugars (Heilbrunn et al., 1958). It is odd that such a small protein has multiple physiologic functions, but this is not too unusual in nature. Heilbrunn et al. noticed that insulin does not directly reduce the amount of glucose in the blood (Heilbrunn et al., 1958). Hormones are small because they must trigger multiple pathways. The hormones themselves are ligands for larger proteins that start certain biological pathways. The small size of insulin and strength if its disulfide bonds allow it to be secreted into the bloodstream without losing too much concentration due to degradation. Insulin binds to the a-subunit of insulin receptor substrate 1 (IRS-1) which causes autophosphorolation in its cytoplasmic B-subunit. Both insulin and the a-subunits of IRS-1 are connected by two disulfide bonds, which help the proteins line up (White et al., 1994). The binding of insulin to IRS-1 starts multiple pathways that involve other proteins seen in Figure 4. If insulin or the a-subunit of IRS-1 is changed or disrupted, the result could cause diabetic symptoms because the accumulation of glycogen in the liver would not be initiated.
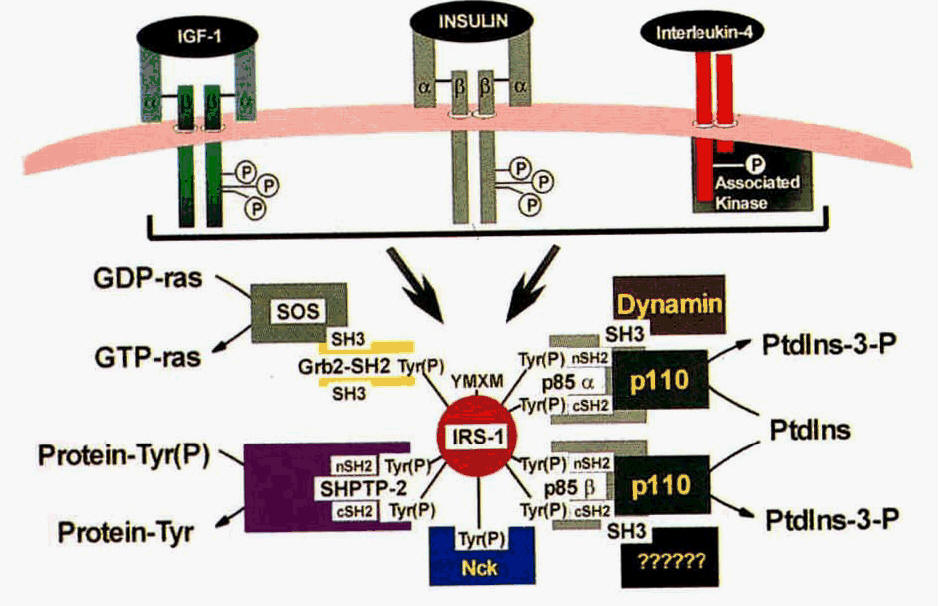
Figure 4. Focus on the upper middle transmembrane protein with insulin bound to it. This is the interaction between insulin and IRS-1 (White et al., 1994).
References
Bowen R. Insulin Synthesis and Secretion. June 15, 1999. <http://www.vivo.colostate.edu/hbooks/pathphys/endocrine/pancreas/insulin.html> last accessed Feb. 15 2010.
Jian Sun X., Rothenberg P., Kah, C., Becker J., Araki E., Wilden P., Cahil D., Goldstein B., White M. Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein. Nature 352, 73-77 (1991).
Heilbrunn L., Ashton F., Feldherr C., Wilson W. The Action of Insulin on Cells and Protoplasm. Biological Bulletin 115. 459-470 (Dec., 1958).
Pfizer Inc. EXUBRA (insulin human [rDNA origen]) Inhalation Powder EXUBRA Inhale. April 2008.< http://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?id=7043&type=display> last accessed Feb. 15 2010.
Roy S., Mukherjee M., Bhattacharya S. Mandal C., Kumar L., Dasgupta S., Bandyopadhyay I., Wakabayashi K. A New Cell Secreting Insulin. Endocrinology 144, 1585-1593 (2003).
Strakoshc C., The Discovery of Insulin. Ramsy Health Care, 1-20 (2005).
White M., Kahn R. The Insulin Signaling System. The Journal for Biological Chemistry 269, 1-4 (1994).
Questions? Contact me at Joholzwarth@davidson.edu
Last updated Feburary 17, 2010
Please direct questions or comments to my email name at davidson dot edu